MRI Innovation Reaches a Major Milestone
Research By: Charles Dumoulin, PhD
Post Date: February 19, 2026 | Publish Date:
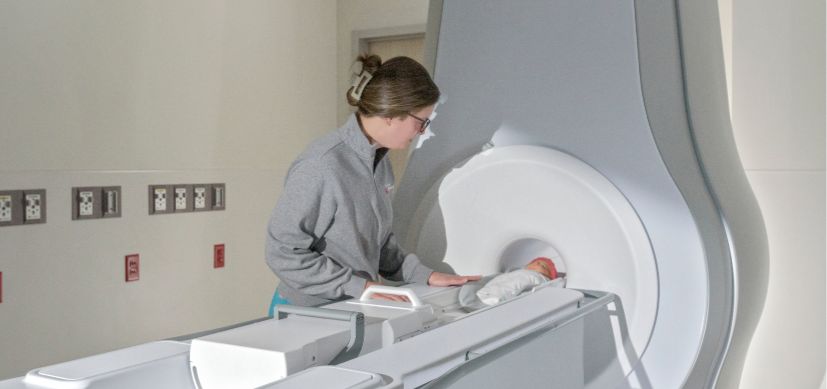
Decades of research have culminated in FDA clearance for a groundbreaking neonatal MRI system designed specifically for infants—reshaping how clinicians diagnose and care for vulnerable newborns.
Cincinnati Children’s scientist Charles Dumoulin, PhD, spent decades researching how to create a safe way to provide high-quality MRI imaging for newborns in the neonatal intensive care unit. Now, an innovation based on that research has reached a major milestone.
The spinout company Eyas Medical Imaging has received U.S. Food and Drug Administration 510(k) clearance for its Ascent3T™ Neonatal Magnetic Resonance Imaging System, the first high-field, 3 Tesla MRI scanner designed specifically for infants. Conceived at Cincinnati Children’s and shaped by Dumoulin’s research and more than 1,700 infant MRI scans performed here on prototype systems, the technology is a breakthrough in neonatal imaging.
Reinventing MRI for the smallest patients
Newborns in the NICU are among the most vulnerable patients in the hospital, but traditional MRI systems were never built with their needs in mind. Transporting critically ill infants from the NICU to distant imaging areas poses serious risks, and adult MRI units often can’t adequately accommodate neonatal anatomy.
Dumoulin, a physicist and professor of Pediatrics and Radiology, set out to change that. His work led to the Ascent3T — a compact MRI system powerful enough to deliver high-resolution 3T imaging while small enough to be installed directly in the NICU. By eliminating transport, clinicians can gather crucial information about vital anatomy, including the brain, lungs, heart and abdomen, right in the NICU.
The system includes thoughtful design elements informed by Cincinnati Children’s early research. A detachable patient table can double as a transport device, supporting varied NICU workflows. And unlike most MRI systems, the Ascent3T is essentially helium free and does not require a quench pipe or external venting, reducing installation complexity and costs for hospitals.
From research to commercialization
Cincinnati Children’s was the first site to install the system for research use. With commercialization support from its Innovation Ventures team, Cincinnati Children’s moved the technology beyond the research environment by licensing the intellectual property to Eyas Medical Imaging, a startup spun out from the health system.
“This is a strong example of how a health system can help move promising technology out of the research setting and into real-world use,” says Nick Archer, vice president of Innovation Ventures. “By partnering early with the inventors and supporting the transition to a startup, we helped ensure the technology was positioned to reach patients and clinicians.”
In 2023, Eyas appointed former Procter & Gamble executive Matt Storer as CEO to guide the company through regulatory steps and early growth. “510(k) clearance marks a significant milestone for the company and reflects our mission to enable healthcare professionals to save more babies’ lives with state-of-the-art, precision imaging,” Storer says.
A milestone for neonatal care
The Ascent3T’s FDA clearance represents an important advance for caregivers seeking safer, better ways to image their smallest patients. “We took great care in the design of the Ascent3T,” Dumoulin says. “Our goal is to transform neonatal care by bringing an unprecedented level of MR imaging and access to the most vulnerable patients when and where they need it.”
Read the full press release
Don’t Miss a Post:
- Subscribe to the Research Horizons Newsletter
- Follow Cincinnati Children’s Research Foundation on Bluesky, X and LinkedIn
Research By

My research interests encompass magnetic resonance imaging (MRI), biomedical engineering and physics. My goals include bringing MRI into the neonatal intensive care unit (NICU) and developing innovative image-guided therapies.