Biomarkers Predict Severe Kidney Injury in Sepsis
Research By: Natalja Stanski, MD | Stuart Goldstein, MD
Post Date: August 6, 2024 | Publish Date: Aug. 6, 2024
Critical Care Medicine | Top Scientific Achievement

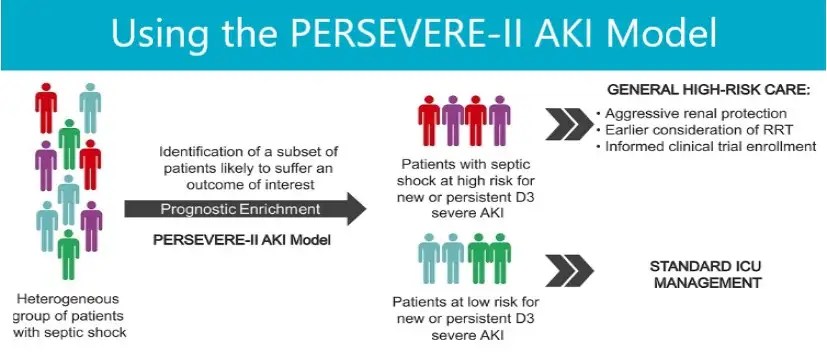
A Cincinnati Children’s–led study validated a powerful new tool for predicting acute kidney injury (AKI) in children with septic shock, a life-threatening condition with high risk for organ failure and death.
The Updated Pediatric Sepsis Biomarker Risk for Acute Kidney Injury (PERSEVERE-II AKI) model—developed using blood-based biomarkers that reflect the body’s immune response—accurately identified children at risk of developing or sustaining severe kidney injury within three days of diagnosis.
This multicenter validation study, published online in August 2024 in Pediatric Critical Care Medicine, involved 363 children treated in pediatric intensive care units across the United States. The model demonstrated an area under the receiver operating characteristic curve (AUROC) of 0.89, a sensitivity of 77%, and a specificity of 88%, outperforming traditional serum creatinine testing. Children predicted by the model to be at high risk were nearly 11 times more likely to develop severe AKI and had a significantly lower likelihood of renal recovery.
The researchers hope to bring this biomarker-based test to the bedside, enabling real-time, precision care for sepsis-associated kidney injury.
“By identifying which patients are most vulnerable within the first day of septic shock, we can target kidney-protective strategies earlier and improve outcomes,” says Natalja Stanski, MD, critical care specialist at Cincinnati Children’s and lead author.
Next steps include integrating PERSEVERE-II into rapid testing platforms and combining it with other validated tools, such as the Renal Angina Index (developed by Stuart Goldstein, MD, at Cincinnati Children’s and recently licensed to RAIDAR Health), to further refine early risk stratification. This approach could ultimately guide enrollment in clinical trials and inform the development of targeted therapies that protect kidney function during sepsis.
About the Study
Cincinnati Children’s co-authors were Bin Zhang, PhD; Mihir Atreya, MD, MPH; Andrew Lautz, MD; Basilia Zingarelli, MD, PhD; Stephen Standage, MD; and Jennifer Kaplan, MD. Collaborating sites included UCSF Benioff Children’s Hospital, Children’s Hospital of Philadelphia, Texas Children’s Hospital, Akron Children’s Hospital, Children’s Hospital of Orange County, Riley Hospital for Children, Children’s Mercy Hospital, Penn State Hershey Children’s Hospital, University of Florida Health Shands Children’s Hospital, Lucile Packard Children’s Hospital Stanford, and Nemours Children’s Health.
Funding sources for this study included the National Institute of General Medical Sciences (K23GM151444-01, K08GM148957-01, L40GM134527-03, R21GM150093-01, R21GM151703, R21GM15009 ), the National Institute of Diabetes and Digestive and Kidney Diseases (K23 DK119463, P50 DK114786), the Eunice Kennedy Shriver National Institute of Child Health and Human Development (R01 HD102396, R01 HD101528), the CDC (75D30123C17693), the Commonwealth of Pennsylvania Department of Health, and the Cincinnati Children’s Research Foundation.
Explore the 2025 Research Annual Report
Don’t Miss a Post:
- Subscribe to the Research Horizons Newsletter
- Follow Cincinnati Children’s Research Foundation on Bluesky, X and LinkedIn
| Original title: | Diagnostic Validation of the Updated Pediatric Sepsis Biomarker Risk (PERSEVERE-II) for Acute Kidney Injury Prediction Model in Pediatric Septic Shock |
| Published in: | Pediatric Critical Care Medicine |
| Publish date: | Aug. 6, 2024 |
Research By


My research is focused on short- and long-term outcomes for children who have AKI and those who are at risk for AKI.







