Four Outstanding Research Achievements of 2025
Post Date: April 30, 2026 | Publish Date:
Research Annual Report 2025

As one of the nation’s leading pediatric research centers, Cincinnati Children’s translates scientific discovery into life-changing innovations that improve child health worldwide. The following four examples from the past fiscal year reflect some of the most impactful research across our health system, spanning the full continuum from discovery science to late-stage clinical trials and system-wide care transformation.
They illustrate this breadth: a Phase 3 clinical trial advancing an inhaled therapy that improves outcomes for patients with a rare and life-threatening lung disease; foundational microbiome and immune research revealing a way to restore infant immune defenses disrupted by early antibiotic exposure; a national implementation science effort embedding emotional health into routine pediatric specialty care; and preclinical innovation using mRNA technology to reprogram immune responses and prevent severe allergic reactions.
Collectively, this work exemplifies a mission-driven research enterprise where discovery is only the beginning, and every breakthrough is focused on improving health outcomes for children and families in Cincinnati and across the globe.
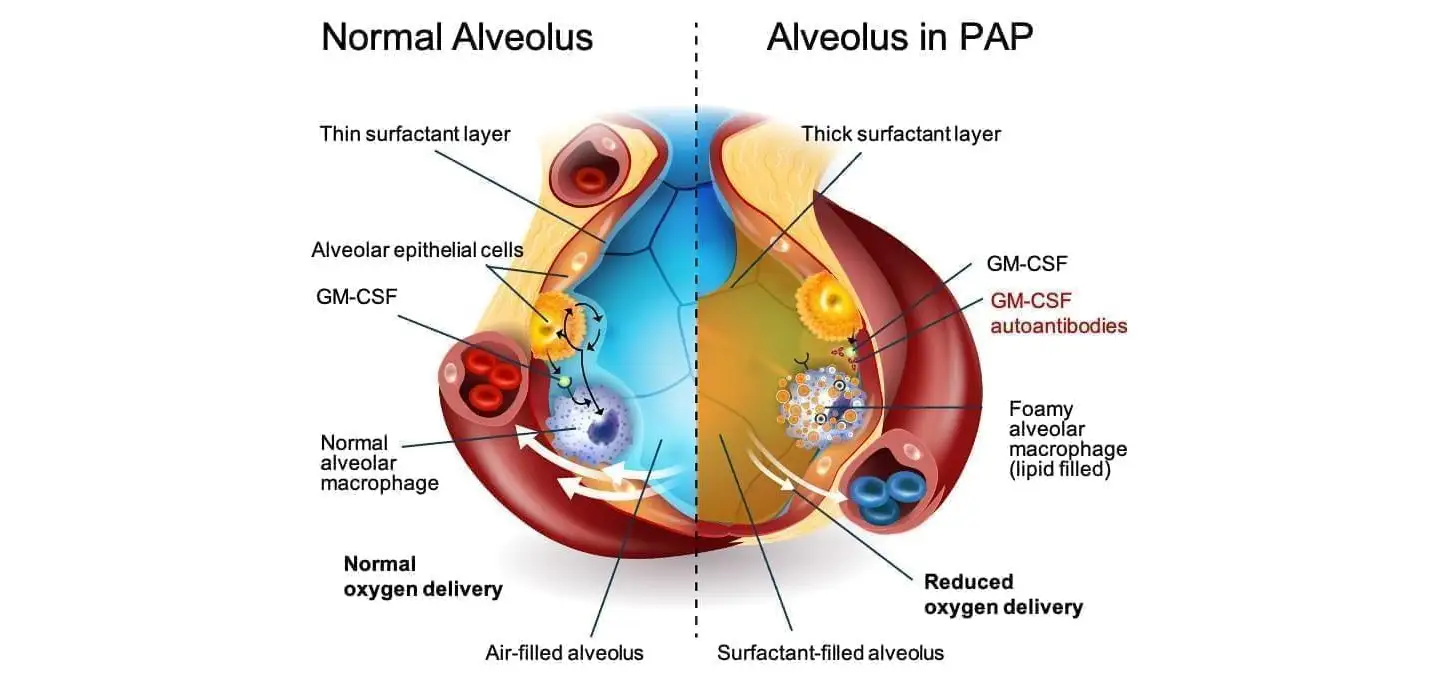
A New Option for a Devastating Lung Disease
For people living with autoimmune pulmonary alveolar proteinosis (aPAP), survival has long depended upon an invasive procedure known as whole‑lung lavage. But investigators at Cincinnati Children’s have been leading a years-long hunt to develop a better way.
That work paid off in August 2025 with the publication of results from a Phase 3 clinical trial led by Bruce Trapnell, MD, director of the Translational Pulmonary Science Center at Cincinnati Children’s. The study, published in The New England Journal of Medicine, evaluated molgramostim, an inhaled form of granulocyte‑macrophage colony‑stimulating factor (GM‑CSF). The IMPALA-2 trial was conducted at 43 sites in 16 countries, including the U.S., Canada, Japan, France, Germany, Italy, Ireland, Romania, Denmark, South Korea, Australia, and Turkey.
In aPAP, patients produce antibodies that block GM‑CSF, preventing lung immune cells from clearing surfactant. The resulting buildup interferes with oxygen exchange and can progress to respiratory failure. After 48 weeks of treatment, participants receiving molgramostim showed significantly greater improvements in lung gas transfer than those receiving placebo. Patients also demonstrated better exercise capacity and reported improvements in respiratory‑related quality of life.
While the therapy did not eliminate the need for whole‑lung lavage, fewer patients required the procedure during the study period. The treatment was generally well tolerated, with no major safety concerns identified.
Data from this clinical trial has been presented to the U.S. FDA for review. In September 2025, the drug maker Savara Inc. presented more data at the ERS Congress 2025 in Amsterdam. If approved, it would complete a nearly three-decade research quest to directly treat the disease versus managing symptoms.
“There are still questions to be answered, including those related to dosing and administration and the time required to achieve maximal therapeutic benefit,” Trapnell says. “We will continue to pursue these questions in our efforts to improve the lives of people living with aPAP.”
Read more about this research
Restoring Infant Immune Defenses After Antibiotics
Antibiotics save lives, but growing evidence shows they can also disrupt immune development when given early in life. In June 2025, Cincinnati Children’s researchers published a study in Cell that not only clarified how this damage occurs, but also pointed to a potential way to reduce the risk.
The research was conducted over four years by MD/PhD students Jake Stevens and Erica Culberson, both Albert B. Sabin Scholars at Cincinnati Children’s, and senior author Hitesh Deshmukh, MD, PhD, a neonatologist with the Perinatal Institute who has since joined the University of Rochester Medical Center.
The research focused on how early‑life antibiotics alter the gut microbiome and, in turn, impair immune defenses in the lungs. Investigators found that infants exposed to antibiotics developed fewer protective CD8⁺ memory T cells—key immune cells that help fight respiratory infections. The effect was observed in both mouse models and human infant samples, and in mice, the immune deficits persisted into adulthood.
Digging deeper, the team identified a critical link between beneficial gut bacteria and lung immunity. They found that Bifidobacterium, a species of beneficial bacteria commonly found in healthy infant guts, produces a molecule called inosine that plays a critical role in immune cell development.
Antibiotic exposure disrupts inosine production. However, when mice received inosine supplements, normal immune development resumed and infection resistance improved.
“These remarkable findings indicate that we might be able to protect at-risk infants through targeted supplementation,” Deshmukh says. “However, it will require more testing and confirmation through human clinical trials before any clinical recommendations could be made.”
Read more about this research
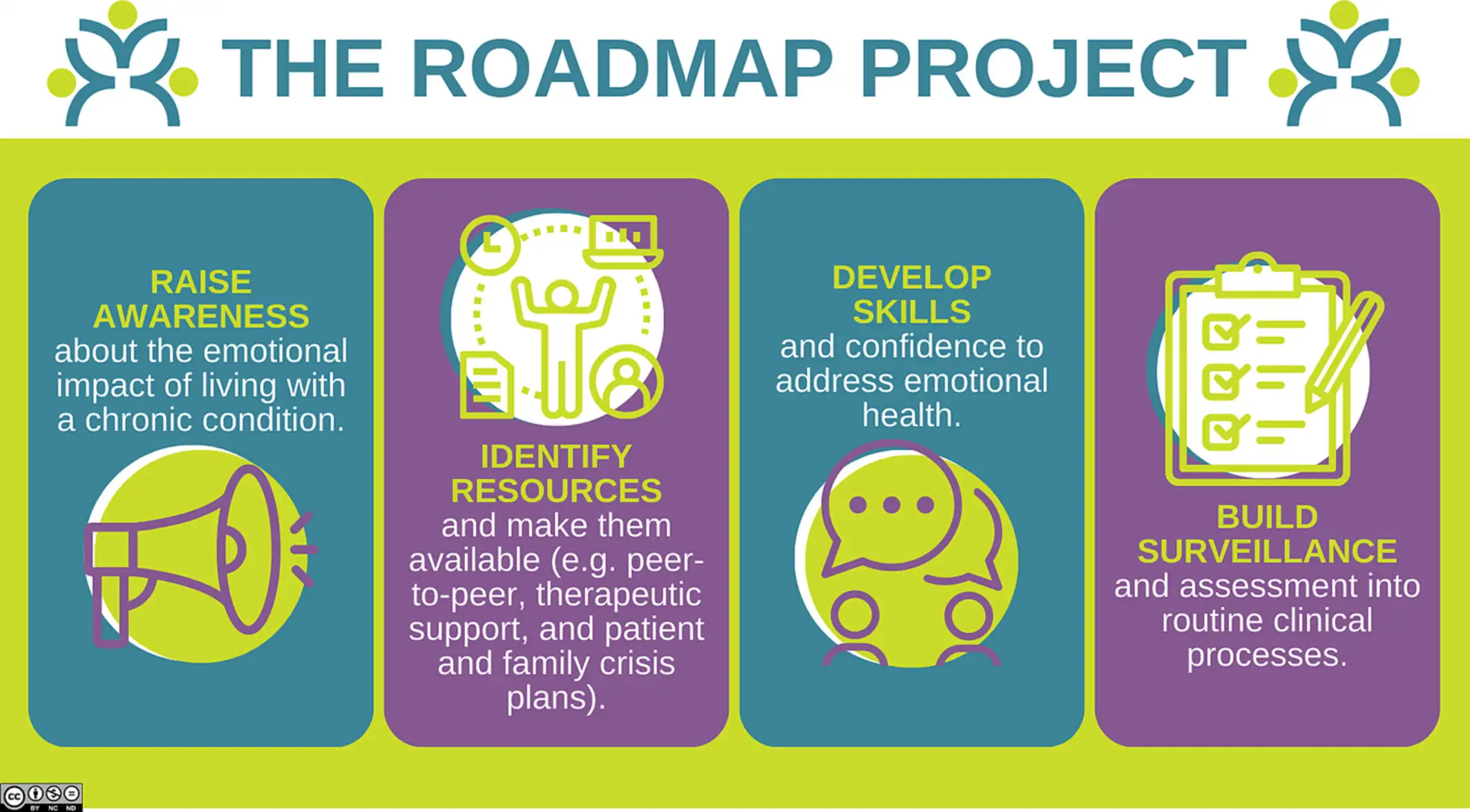
Making Emotional Health Part of Everyday Pediatric Care
As pediatric mental health needs continue to rise, a team of Cincinnati Children’s investigators and partners has focused on a practical question: how can emotional health be addressed routinely in specialty care settings, especially for children with chronic medical conditions?
A June 2025 publication in Translational Behavioral Medicine describes a national effort to do just that. The research was led by first author Jill M Plevinsky, PhD, from Children’s Hospital of Philadelphia and senior author Lori Crosby, PsyD, from Cincinnati Children’s.
The study outlines the Roadmap for Emotional Health Project, co‑led by the American Board of Pediatrics and James M. Anderson Center for Health Systems Excellence at Cincinnati Children’s. Carole Lannon, MD, MPH, is the Roadmap project leaders and served as a co-author on this study.
Rather than the existing practice of shuttling children between systems, the Roadmap emphasizes integrating emotional health into existing pediatric specialty care practices, with pediatric psychologists playing key system‑level roles.
The Roadmap has been piloted successfully in 58 clinical teams across 15 subspecialties from 35 children’s hospital systems. Central to the approach: the Normalize–Ask–Pause–Connect (N‑A‑P‑C) framework, which provides a structure for clinicians to acknowledging emotional challenges, invite conversation, and connect families with support.
“The value of psychology colleagues supporting clinical teams in addressing patient and family emotional health is clear,” Crosby says. “Better integration of emotional and physical health can improve patient experiences, prevent behavioral escalations, and help to reduce long-term healthcare concerns. This work reflects a shift toward whole‑child care, where emotional well‑being is recognized as inseparable from physical health.”
This research is one of many improvement outcomes flowing from a major expansion and restructuring of mental and behavioral health services and science at Cincinnati Children’s. Learn more about our Mental and Behavioral Health Institute.
Read more about this Roadmap

Reimagining Allergy Treatment With mRNA Technology
Allergic diseases affect millions of children and adults, yet treatment often focuses on managing symptoms rather than preventing reactions altogether. In September 2025, a preclinical study published in the Journal of Clinical Investigation, described a novel vaccine‑style approach that could change that paradigm.
The study was led by Marc Rothenberg, MD, PhD, director of the Division of Allergy and Immunology at Cincinnati Children’s and Drew Weissman, MD, PhD, a scientist at the University of Pennsylvania who was awarded a Nobel Prize in 2023 for his contributions to the mRNA technology that led to the first effective COVID vaccines.
The new project evaluated an allergen‑specific mRNA vaccine delivered via lipid nanoparticles. In mouse models of severe allergy, the approach reduced inflammatory immune responses and protected against allergic reactions after exposure to allergens.
Allergic diseases affect nearly 30% of the global population, with food allergy affecting 10% of the population. In addition to high-cost hospital care, lost work days and disrupted schooling, about 250,000 people a year die from asthma—a dangerous outcome of allergic reactions. All of which point to a need for improved prevention.
Rather than broadly suppressing the immune system, the mRNA vaccine strategy aims to retrain the system, promoting tolerance to specific allergens. Because many allergic diseases share common inflammatory pathways, researchers say the vaccine platform could be adapted to multiple forms of allergy.
“These exciting findings have the potential to be a revolutionary approach for allergy interventions,” Rothenberg says.
Eventually, mRNA vaccines could offer an alternative to long‑term allergy shots and often hard-to-follow avoidance strategies. But first, a series of clinical trials would be needed to demonstrate safety and effectiveness.
Read more about this research
Explore the 2025 Research Annual Report







