$10.3M NIH HEAL Grant Powers a New Era in Pediatric Chronic Pain Research
Research By: Susmita Kashikar-Zuck, PhD | Michael Jankowski, PhD | Christopher King, PhD
Post Date: March 20, 2026 | Publish Date:

Cincinnati Children’s team will map the biology behind persistent pain to unlock better therapies
Chronic pain affects millions of children and teens, disrupting school, sleep, sports, friendships, and mental health. Yet even as pediatric pain clinics have expanded, families still ask a question science has struggled to answer: Why does pain continue when the injury or illness is gone?
The team behind a new five‑year, $10.3 million NIH HEAL Initiative grant awarded to Cincinnati Children’s aims to find the answer. The project brings together a multidisciplinary team—clinicians, neuroscientists, psychologists, immunologists, and basic scientists—to uncover the biological roots of chronic pain in children and translate those insights into more effective, personalized treatments. Susmita Kashikar‑Zuck, PhD and Christopher King, PhD, of the Division of Behavioral Medicine and Clinical Psychology and Michael Jankowski, PhD, of the Department of Anesthesia are teaming up to lead the grant.
“We’ve made progress helping patients cope and function,” says clinical psychologist Kashikar-Zuck, associate director of the Pediatric Pain Research Center and a founding member of the Chronic Pain Center at Cincinnati Children’s. “Now we’re going after the ‘why’—the biology driving persistent pain—so we can prevent it and treat it more effectively.”
Why This Matters Now
Chronic pain in children can arise from a known medical condition or with no clear cause at all. For many, imaging and lab tests show “normal” results, leaving families without biological explanations for very real symptoms. Researchers still don’t know what biological processes—across the brain, spinal cord and immune system—drive these persistent symptoms. This project will investigate mechanisms such as inflammation, stress‑responsiveness, altered communication between neurons and immune cells, and early‑life sensory neuron priming to uncover why pain becomes chronic in some children.
“If we want to truly help kids thrive, we need a deep understanding of the biological processes taking shape during development,” says King, associate professor of Pediatrics. “This insight will allow us to move toward more effective and targeted treatments.”
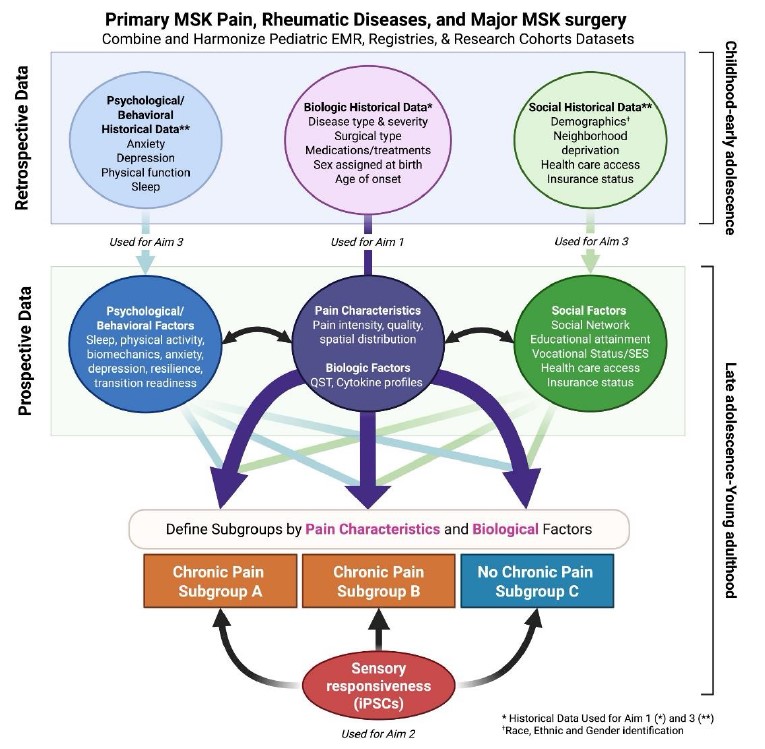
A Comprehensive Look at Pain—from Symptoms to Cells
The project takes a uniquely integrated approach, studying each child through multiple lenses:
- Pain sensitivity: The team will use quantitative sensory testing (QST) to measure how children respond to temperature and pressure, helping identify sensory patterns that may be linked to persistent pain.
- Immune and inflammatory signatures: Blood samples will undergo cytokine profiling to map immune pathways that may contribute to ongoing pain.
- Neuro‑immune signaling in sensory neurons: Using patients’ serum in induced pluripotent stem cell (iPSC)–derived sensory neuron models, researchers will study how immune signals influence neuronal activity involved in pain.
- Integrated clinical data: Retrospective registry and clinical records—combined with prospective pain and symptom measures—will create a rich dataset connecting lab findings with key psychosocial influences and day‑to‑day functioning.
- Mechanistically distinct subgroups: Model‑based, unsupervised clustering approaches will be used to identify biological “biotypes” of chronic musculoskeletal pain, paving the way for targeted interventions.
Together, these data will allow investigators to uncover pain subtypes—subgroups of children who share similar biological patterns and risk factors. Those subtypes can then guide the development of targeted therapies, rather than relying on trial‑and‑error treatment.
From Discovery to Better Treatments
A key strength of the program is its tight link between clinical research and basic science.
“When we see a feature in the clinic—like heightened pain sensitivity or evidence of immune activation—we can test these mechanisms directly in our lab models,” says Jankowski, vice chief for research in the Department of Anesthesia. “When clinical and basic science findings converge, that’s when we start uncovering novel therapies.”
The long‑term vision is to move beyond one‑size‑fits‑all approaches to pain management and toward precision treatments tailored to a child’s biology. That could include targeted behavioral strategies, non‑pharmacologic approaches, or new medications guided by the pathways the team uncovers.
What’s Ahead
Over the next five years, the team will: build a deeply characterized pediatric cohort, identify biological drivers of persistent pain, use machine learning to define diagnostic biotypes, and pilot mechanism‑guided therapies based on those findings.
It’s an ambitious effort—but one with the potential to reshape pediatric pain care.
“This grant allows us to connect what kids feel with what cells are doing,” Kashikar-Zuck says. “That’s the first step toward giving them better days, better function, and the chance to reach their full potential.”
-
Read the project details
-
Learn more about the Division of Behavioral Medicine and Clinical Psychology
-
Learn more about the Division of Anesthesia
Don’t Miss a Post:
- Subscribe to the Research Horizons Newsletter
- Follow Cincinnati Children’s Research Foundation on Bluesky, X and LinkedIn
Research By


Our lab is interested in the mechanisms of sensory neuron sensitization with a particular emphasis on nociceptive processing.