TSPAN12 Gene Plays Central Role in Fibrosis Development Among Some People with EoE
Research By: Tetsuo Shoda, MD, PhD | Marc Rothenberg, MD, PhD
Post Date: October 20, 2021 | Publish Date: Oct. 20, 2021
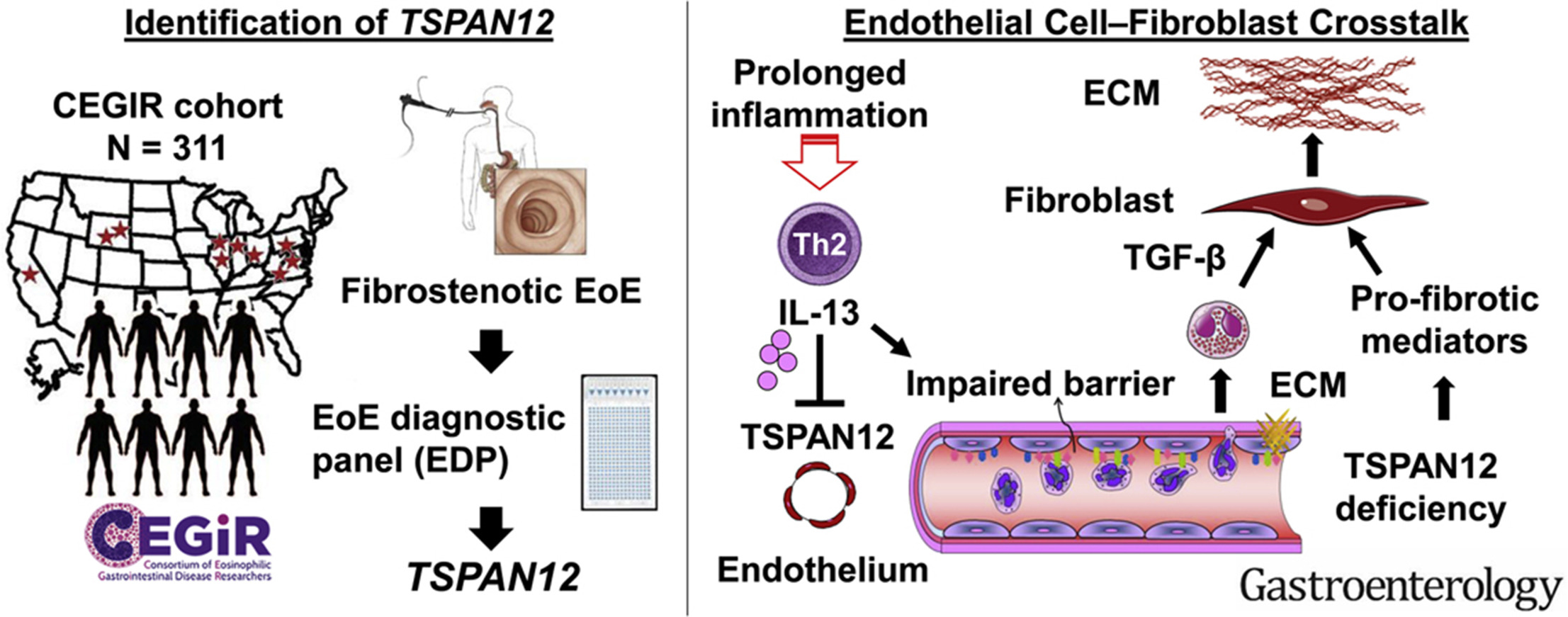
When the gene TSPAN12 malfunctions, people with eosinophilic esophagitis (EoE) appear more likely to develop a serious form of scarring in their esophagus that can make it much harder for them to swallow food.
This new finding, published Oct. 20, 2021, in Gastroenterology, comes from a 16-center consortium of EoE experts based at Cincinnati Children’s and led by Marc Rothenberg, MD, PhD, Director, Division of Allergy and Immunology. Tetsuo Shoda, MD, PhD. was first author.
The TSPAN12 gene, which helps regulate cell development, growth and motility, is found in vascular endothelial cells in the esophagus. This gene and cell type had not previously been identified as having an important role in the fibrosis associated with EoE. Now that a role appears likely, the new findings suggest that a different course of treatment may be needed for this subset of patients with EoE.
What is EoE?
Eosinophilic esophagitis is a severe form of food allergy that occurs when the body reacts to food allergens by flooding the esophagus with eosinophils, a type of white blood cell produced by the immune system.
The resulting waves of tissue inflammation can interfere with swallowing. To control them, clinicians may prescribe topical and systemic steroids, dietary restrictions often involving multiple food groups, and/or proton-pump inhibitors. More recently, medications that target specific molecular pathways active in EoE have been tested in clinical trials.
What is fibrostenosis?
Fibrostenosis is a build-up of scar-like rings and strictures often seen occurring in the intestine as a complication of Crohn’s disease. Long-term inflammation triggered by excess levels of eosinophils can cause a similar type of damage in the esophagus.
Only some people with EoE develop fibrostenosis. Sometimes the condition emerges at an early age, while others experience the damage slowly building over many years. The development of fibrostenosis may represent one or more distinct phenotypes, but so far, experts have not been able to predict which patients with EoE are most at risk.
How the TSPAN12 findings may influence treatment
Topical steroids that often are used effectively to treat EoE have variablevalue against fibrostenosis. However, it may be possible to normalize TSPAN12 levels with anti–interleukin 13 therapy, the co-authors say.
This study involved analyzing biopsy tissues from 394 children and adults, including 311 people diagnosed with EoE.
At the time of biopsy, 72% of patients with EoE were taking either steroids or proton pump inhibitors, and 47% were following diet therapy. The study found that about half of the patients with EoE had fibrostenotic rings or strictures. The fibrostenosis was most common among adult females and less common among those on elimination diets.
Genetic analysis revealed that TSPAN12 was the most common of several genes whose expression differed in association with fibrostenosis. Single-cell RNA sequencing revealed that TSPAN12 expression was substantially decreased in inflamed endothelial tissue in the esophagus in patients with active EoE.
The team also reports that the lack of TSPAN12 in endothelial cells promotes the tissue remodeling that may lead to the formation of rings and strictures.
In lab tests, the team also found that that anti–interleukin 13 antibody (QAX576) treatment increased TSPAN12 expression.
Next steps
Further study is needed to replicate elements of these findings, including prospective studies evaluating anti–interleukin 13 treatment and whether TGF-β could be used to moderate the downstream effects of TSPAN12 deficiency.
About this study
Funding sources for this work includes the Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR), a part of the NIH-funded Rare Diseases Clinical Research Network (U54 AI117804). Cincinnati Children’s hosts CEGIR and serves as the data management and coordinating center for the RDCRN.
CEGIR also is supported by the American Partnership for Eosinophilic Disorders (APFED), Campaign Urging Research for Eosinophilic Disease (CURED), and Eosinophilic Family Coalition (EFC).
| Original title: | Loss of Endothelial TSPAN12 Promotes Fibrostenotic Eosinophilic Esophagitis via Endothelial Cell–Fibroblast Crosstalk |
| Published in: | Gastroenterology |
| Publish date: | Oct. 20, 2021 |
Research By








