Roots of New Drug for HES Trace Back to Cincinnati Children’s
Post Date: September 28, 2020 | Publish Date:
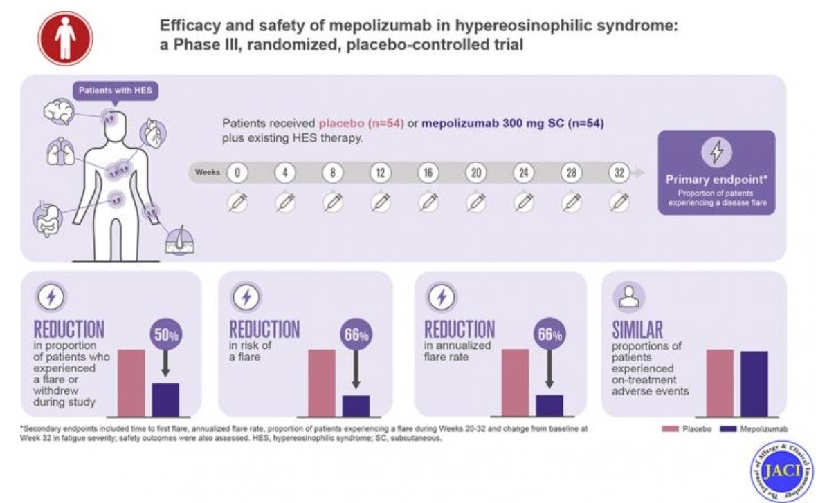

In 2008, Marc Rothenberg, MD, PhD, Director of the Division of Allergy and Immunology and the Cincinnati Center for Eosinophilic Disorders at Cincinnati Children’s, led an international group of investigators to conduct a randomized clinical trial that proved the ability of mepolizumab to lower oral steroid doses in patients with HES.






