Research Reveals Potential Treatment to Prevent Obesity-Driven Liver Damage
Research By: Maria E. Moreno-Fernandez, PhD | Senad Divanovic, PhD
Post Date: May 17, 2021 | Publish Date: May 17, 2021
Team led by experts at Cincinnati Children’s reports success– in mice–at controlling a subgroup of immune system cells that trigger non-alcoholic fatty liver disease (NAFLD)
One of the especially dangerous health risks of being extremely overweight occurs when an obese person begins to accumulate fat in their liver.
This condition—non-alcoholic fatty liver disease (NAFLD)—is the world’s most common chronic liver disease and is the primary underlying cause for liver transplants in children and adults. Without such transplants, which are available to only a small percentage of patients, NAFLD over time can be fatal. In fact, (excluding alcohol-related liver damage) more than 30,000 people a year die from NAFLD.
For years, the primary way to treat NAFLD has been through the use of various weight control methods: diet programs, exercise regimens, medications of limited benefit, bariatric surgery, and more. But once people develop progressive NAFLD, simply losing weight is not enough.
Now, after years of studying the numerous mechanisms involved with obesity and NAFLD, a team of 20 scientists at Cincinnati Children’s reports taking a significant step forward. Their findings were published online May 17, 2021, in Cell Metabolism.
Introducing ihTh17 cells
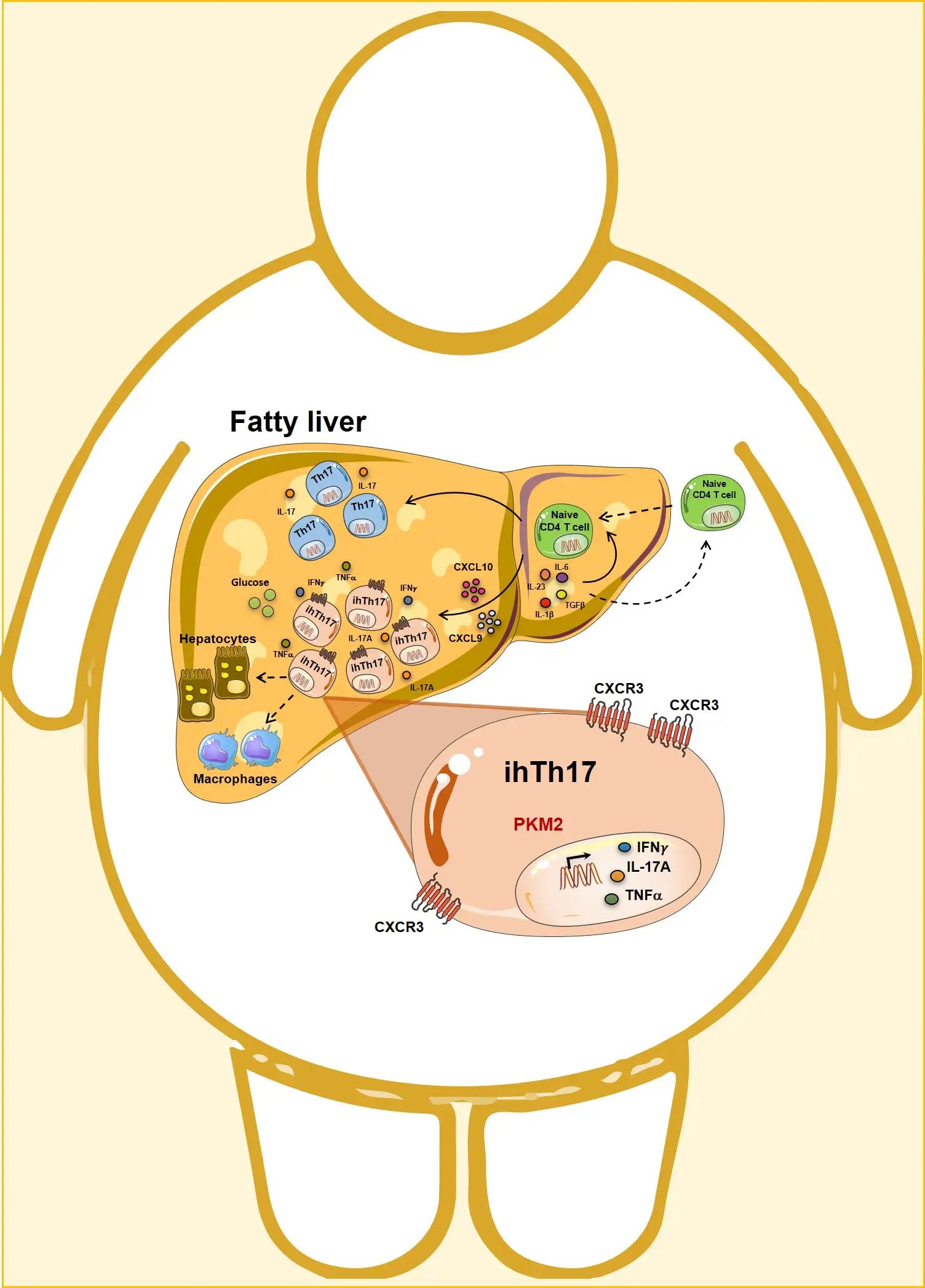
The research team reports that excessive fat deposition in the liver due to obesity can alter the microenvironment of the liver in a way that attracts a highly specific population of immune T cells to the liver. These “inflammatory hepatic CXCR3+Th17 cells” or “ihTh17” cells go on to trigger excess inflammation and life-threatening liver damage.
By running a series of experiments using human tissues and cells and multiple lines of genetically modified mice, the team found that obesity itself triggers activity along a molecular “pathway” that starts with excess expression of the CXCL10 and CXCR3 genes. This abnormal activity attracts more and more ihTh17 cells to the liver. The consequence being a scorched earth inflammatory feedback loop that recruits additional immune cells and progressively damages liver function.
After tracing the ihTh17 cell liver recruitment pathway, the team set out to find a way to break the unhealthy cycle of inflammation. They found success with mice bred to lack expression of the gene Pkm2 in their T cells, which appears to be crucial to continued activity along the CXCR3 pathway.
When these modified mice were given obesity-inducing diets, they still got fat. But they suffered notably less liver damage than non-modified mice.
Next, the researchers tested human tissues collected from people with NAFLD. They confirmed that many of the key genes and molecular activities occurring in the mice also could be detected in the human liver cells.
“Our results demonstrate for the first time that ihTh17 cells represent an important component of the complex world of NAFLD pathogenesis,” say corresponding author Senad Divanovic, PhD, a member of the Division of Immunobiology at Cincinnati and first author Maria Moreno-Fernandez, PhD, a postdoctoral fellow in the Divanovic laboratory.
Learning more about how to regulate ihTh17 cells’ function and their interaction with the liver cells and the immune system could lead to new therapies to reduce the harm caused by NAFLD.
Next steps
But will the treatment approach used in mice also help people? Human gene editing is not likely to be an acceptable option for this condition anytime soon. However, some drugs are known to be capable of blocking Pkm2 activity, Divanovic says.
Those drugs still require more in-depth laboratory evaluation. Ultimately, a promising compound also would need to be tested in multi-year clinical trials. But now, for the first time in years, the team has a promising lead to explore.
“If we can modulate the unwanted inflammatory responses associated with NAFLD in a targeted way we may be able to ameliorate the liver damage and improve the survival and health of people with NAFLD,” Divanovic says.
About the study
These findings reflect the latest insights from scientists, surgeons and clinicians at Cincinnati Children’s who have devoted many years to obesity research. Their previous work has increased understanding of how obesity changes metabolic activity, immune system function, heart health and more. Several of the co-authors on this publication previously helped establishing bariatric surgery as a safe option for obese children that also helps reverse type 2 diabetes.
In addition to Divanovic, co-authors from 15 research divisions and services at Cincinnati Children’s contributed to this study. They include Maria E. Moreno-Fernandez (n.e Fields) (lead author- Division of Immunobiology); Daniel Giles (now at Janssen); Jarren Oates (Division of Immunobiology), Calvin Chan (Division of Immunobiology), Michelle Damen (Division of Immunobiology), Jessica Doll (Division of Immunobiology), Traci Stankiewicz (Division of Immunobiology), Xiaoting Chen (CAGE), Kashish Chetal (Division of Biomedical Informatics), Rebekah Karns (Division of Gastroenterology, Hepatology and Nutrition), Matthew Weirauch (CAGE), Lindsey Romick-Rosendale (NMR Metabolomics Core), Stavra Xanthakos (Division of Gastroenterology, Hepatology and Nutrition), Rachel Sheridan (now at Dayton Children’s), Sara Szabo (Division of Pathology and Laboratory Medicine), Amy Shah (Division of Endocrinology), Michael Helmrath (CuSTOM), Hitesh Deshmukh (Division of Neonatology and Pulmonary Biology), and Nathan Salomonis (Division of Biomedical Informatics). Co-authors also include Thomas Inge (now at Children’s Hospital Colorado).
Funding for this work includes grants from the National Institutes of Health R01DK099222, R01DK099222-02S1, T32AI118697, T32GM063483-14, R01HL142708-01 and P30 DK078392 of the Digestive Disease Research Core Center; the Department of Defense W81XWH2010392; the American Diabetes Association 1-18-IBS-100 and 1-19-PMF-019; the American Heart Association 17POST33650045; and internal funding support from Cincinnati Children’s and the University of Cincinnati.
| Original title: | PKM2-dependent metabolic skewing of an inflammatory hepatic Th17 subset regulates pathogenesis of non-alcoholic fatty liver disease |
| Published in: | Cell Metabolism |
| Publish date: | May 17, 2021 |
Research By