Kidney Injury Device Fares Well in Post-Approval Tracking Study
Research By: Stuart Goldstein, MD
Post Date: February 9, 2026 | Publish Date: Feb. 6, 2026
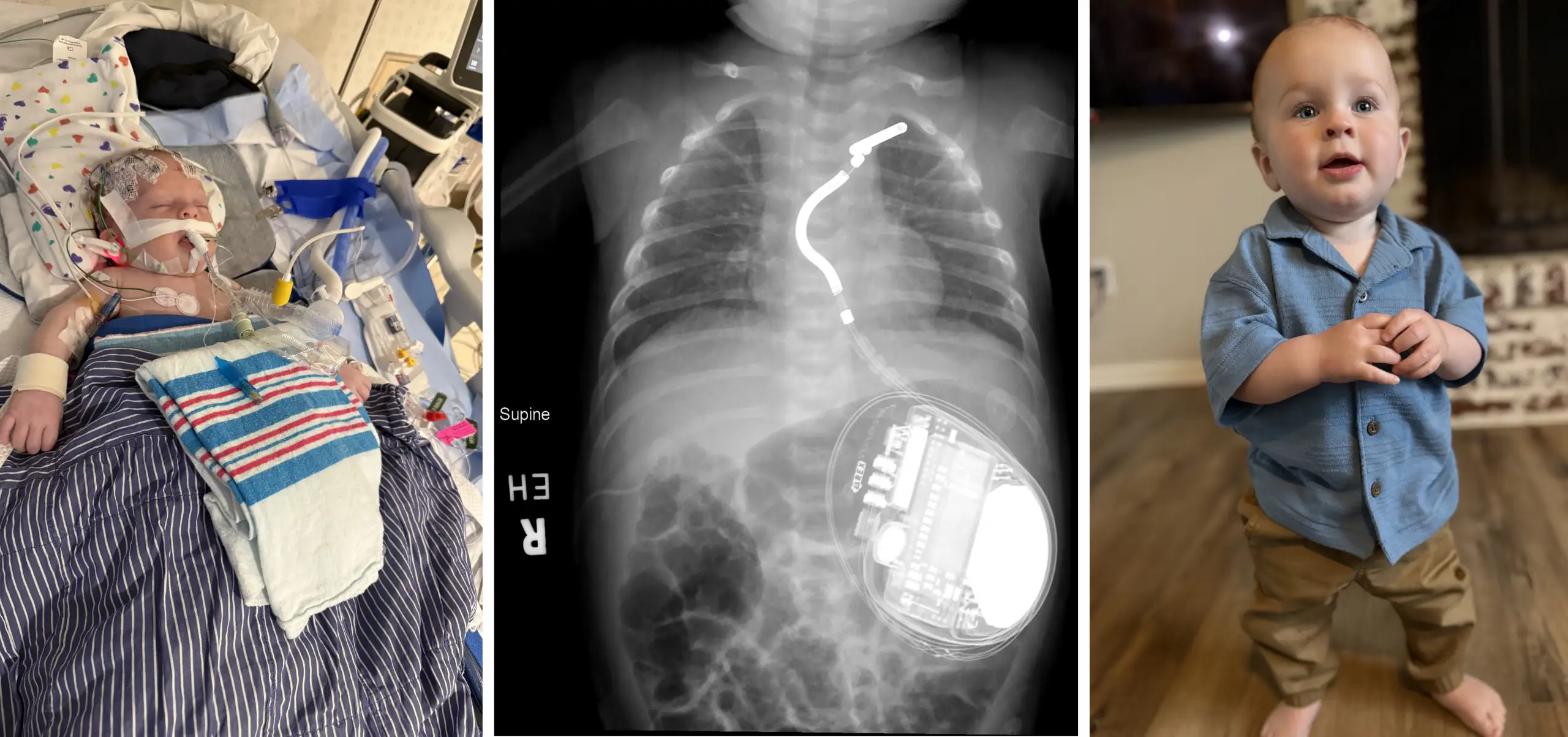
A new device to help children who have life-threatening acute kidney injury (AKI) and sepsis continues showing positive results since its initial FDA approval in February 2024.
Details from a post-approval surveillance registry were published Feb. 6, 2026, in Pediatric Nephrology. Stuart Goldstein, MD, director of the Center for Acute Care Nephrology at Cincinnati Children’s, was the corresponding author.
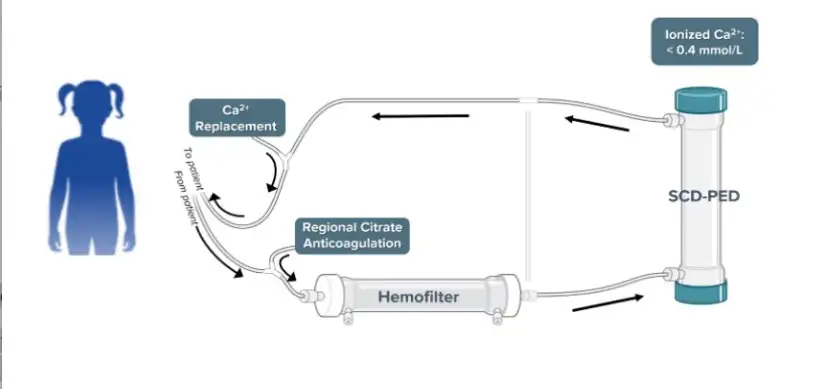
The QUELIMMUNE device, also known as the Selective Cytopheretic Device for Pediatrics (SCD-PED), is connected to existing hemodialysis delivery systems to deactivate neutrophils that can trigger hyperinflammation. The device is made by SeaStar Medical Holding Corporation, which funded the post-approval surveillance study. Goldstein is a member of the SeaStar Medical Scientific Advisory Board.
“These real-world experiences of the QUELIMMUNE therapy are compelling for a number of reasons,” Goldstein says.
“First, they mirror the results from the two registrational trials that enabled FDA approval of the therapy in critically ill patients with AKI and sepsis requiring renal replacement therapy, further demonstrating the reproducibility of the therapy benefit.
“Second, the complexity of the patient population with respect to their comorbidities show a much broader array of use compared to the narrower patient selection in the registrational trials.
“And finally, with no device-related serious adverse events to date, I believe there is a clear potential benefit from the QUELIMMUNE therapy with minimal downside risk to patient safety. For these reasons, we have had the QUELIMMUNE therapy available since FDA approval for our patients who present with such life-threatening conditions.”
Read the full announcement from SeaStar
Read news story in Healio: Nephrology News & Issues
As a member of the SeaStar Medical Scientific Advisory Board, Goldstein has received financial compensation for his service in this capacity.
Don’t Miss a Post:
- Subscribe to the Research Horizons Newsletter
- Follow Cincinnati Children’s Research Foundation on Bluesky, X and LinkedIn
| Original title: | Early post-approval experience of the selective cytopheretic device surveillance registry for pediatric AKI requiring kidney replacement therapy |
| Published in: | Pediatric Nephrology |
| Publish date: | Feb. 6, 2026 |
Research By

My research is focused on short- and long-term outcomes for children who have AKI and those who are at risk for AKI.