Xenon MRI launches at Cincinnati Children’s
Research By: Jason Woods, PhD
Post Date: May 12, 2023 | Publish Date:
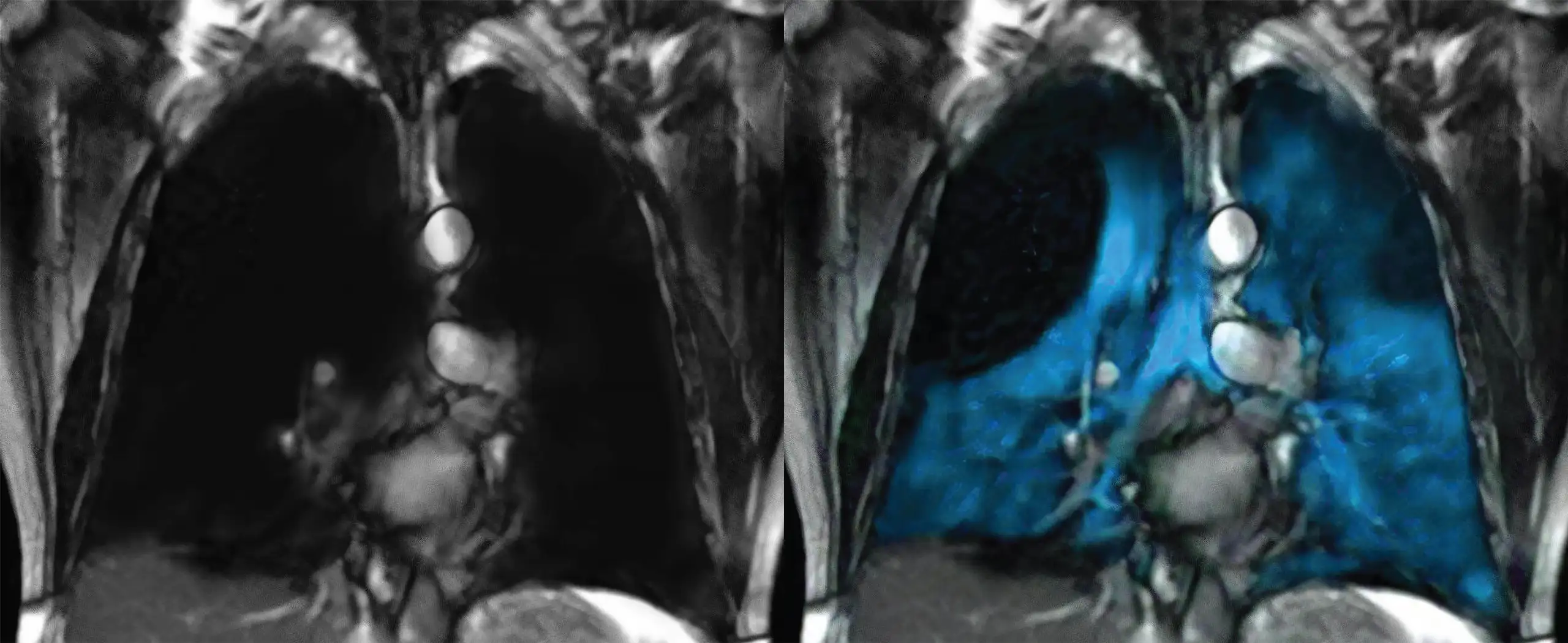
Cincinnati Children’s has become the first hospital in the nation to image a patient with XENOVIEWTM, a novel xenon (Xe) gas blend that enables a safe and detailed view of lung ventilation, since its FDA approval for ages 12 and older in December of 2022.
Jason Woods, PhD, director of research in the division of Pulmonary Medicine at Cincinnati Children’s, has been investigating a way to alleviate these concerns. For more than 20 years, he has researched Xe gas to pioneer a safer, faster, and more precise method for pinpointing lung disease. This research culminated in the launch of the Xenon-129 MRI Clinical Trials Consortium in 2015 which began exploring the potential of hyperpolarized Xe gas imaged via radiation-free magnetic resonance imaging (MRI) as a standardized tool at other sites.
Research By

The primary research areas of the Woods Lab include pulmonary understanding structure-function relationships in lung disease with a focus on cystic fibrosis and neonatal lung diseases via translational research and multi-site clinical trials.







