Vitamin A Helps Our Immune Systems Thanks in Part to Gut Bacteria That Make Their Own Supply
Research By: Vivienne Woo, PhD | Theresa Alenghat, VMD, PhD
Post Date: October 22, 2021 | Publish Date: Oct. 22, 2021
Experts at Cincinnati Children’s discover another way that a healthy microbiome can support human health
The medical community has known for years that vitamin A is a necessary element of a healthy diet. But until now, scientists did not know that mammals need help from the bacteria living in their gut to assure that the vitamin can do its job.
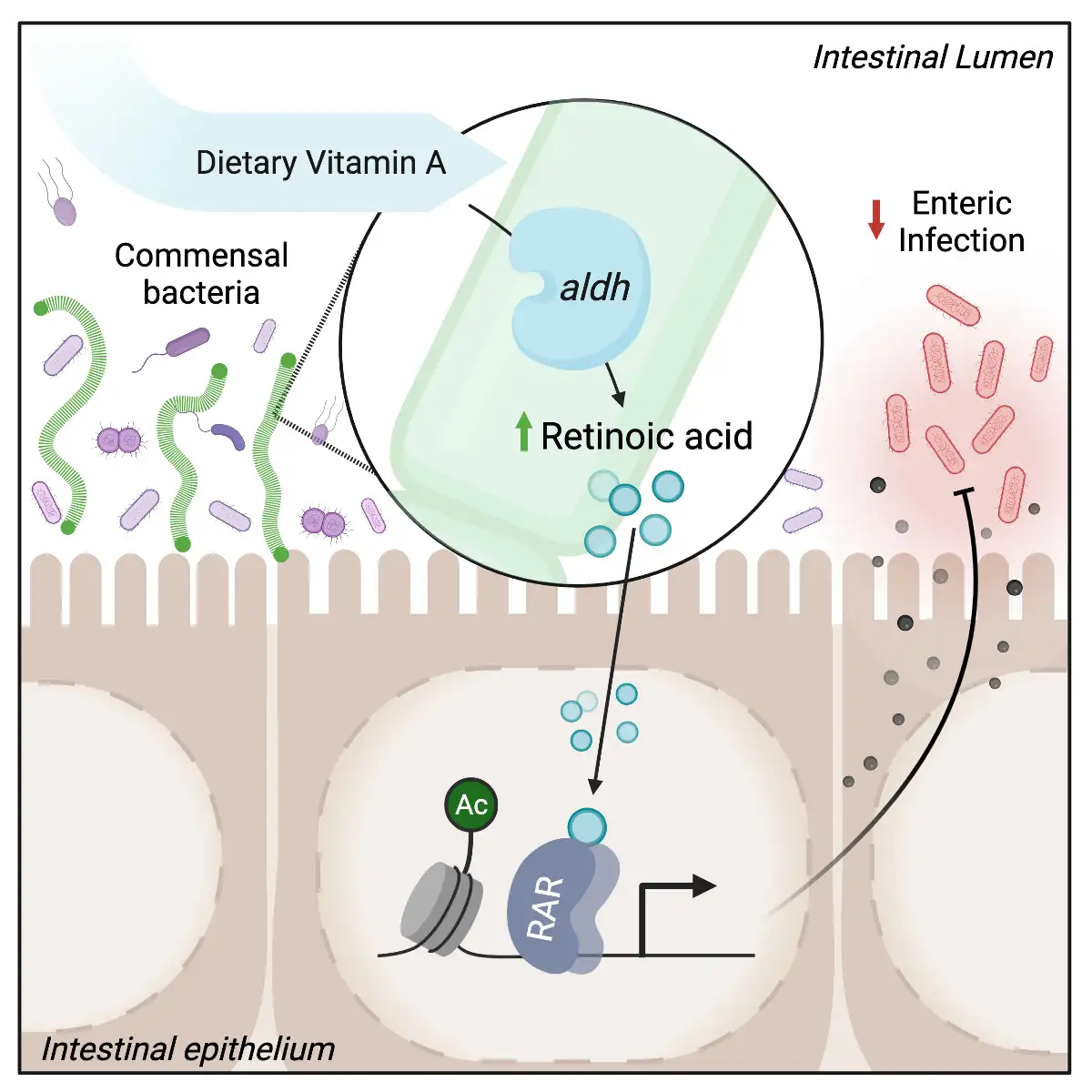
In findings published online Oct. 20, 2021, in Cell Host & Microbe, researchers at Cincinnati Children’s report that commensal or “good” bacteria can generate retinoic acid from vitamin A and this process plays an important role in priming our immune system to fight off gut infections, such as from harmful bacterial strains.
“To the best of our knowledge, the ability of commensal bacteria to provide a direct source of retinoic acid in the intestine that physiologically regulates the mammalian host has not been described,” says microbiota expert Theresa Alenghat, VMD, PhD, Division of Immunobiology and corresponding author on the study.
The study, led by first author Vivienne Woo, PhD, and co-authored by several other scientists at Cincinnati Children’s, suggests that targeted microbiome-based treatment strategies eventually may reduce infections by helping commensal bacteria maintain proper levels of retinoic acid.
“This finding is exciting because it reveals a new level of understanding for how the microbiome can instruct mammalian cells to protect against infection,” Woo says.
Learning more about the microbiome
The study involved a number of experiments involving mice that were exposed to C. rodentium, a model pathogen that mimics disease caused by infectious E. coli in people. While some strains of E.coli are harmless, others can cause severe diarrhea, cramps, kidney failure and, in rare cases, death.
The team determined that a particular strain of commensal (good) bacteria called segmented filamentous bacteria (SFB) produce an enzyme that directly generates retinoic acid in a region of the intestine upstream from where C. rodentium colonizes. This occurs separately from the retinoic acid produced by the animal’s own cells and is important for defending against C. rodentium infection.
This increased retinoic acid from SFB “primes” defense processes within the lining of the intestine called the epithelium. The epithelium can then produce molecules that block pathogens directly or activate the immune system to respond to harmful invaders.
Importantly, when the commensal bacteria failed to produce their own retinoic acid, mice were unable to compensate from the other sources and their epithelial defense mechanisms were less successful at battling infection.
Next steps for this research
Vitamin A and beta-carotene are precursor forms of retinoic acid that are available as a dietary supplements and occur naturally in many foods, such as carrots, spinach, dairy products and liver. Insufficient intake of vitamin A is linked to a number of diseases and conditions including increased susceptibility to infection.
The USDA recommended daily amount of vitamin A is 900 micrograms (mcg) for adult men and 700 mcg for adult women. The new study does not suggest any changes are needed in these recommendations, rather that retinoic acid-producing bacteria could eventually be evaluated as a probiotic strategy in patients.
The new findings remain too early-stage for people to attempt to manipulate their vitamin A intake as a way to prevent illness. More studies are needed to confirm how microbiome-vitamin A interactions work in humans, what situations and timepoints would benefit from intervention, and which types of probiotics would be most effective.
About this study
Other co-authors from Cincinnati Children’s involved in the study include Emily Eshleman, Seika-Hashimoto-Hill, Jordan Whitt, Shu-en Wu, Laura Engleman, Taylor Rice, Rebekah Karns, Joseph Qualls, and David Haslam.
Funding support for the research came from the National Institutes of Health (DK114123, DK116868, F32AI147591, P30DK078392), the Pew Charitable Trust, the Kenneth Rainin Foundation, the Burroughs Wellcome Fund, and the Cardell Fellowship Program.
| Original title: | Commensal segmented filamentous bacteria=derived retinoic acid primes host defense to intestinal infection |
| Published in: | Cell Host & Microbe |
| Publish date: | Oct. 22, 2021 |
Research By