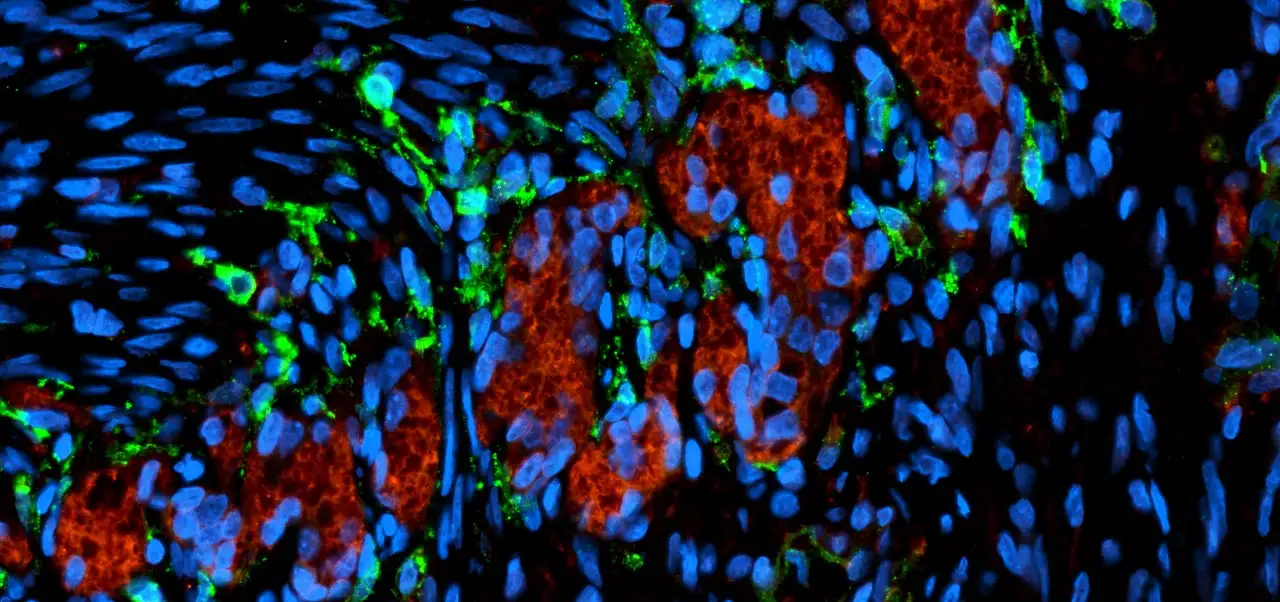
U.S.-Japan Science Team Publishes New Recipe for Creating Complex Organoids
Research By: Keishi Kishimoto, PhD | Aaron Zorn, PhD
Post Date: August 17, 2022 | Publish Date: Aug. 17, 2022
Improved process, developed at Cincinnati Children’s and the RIKEN Center for Biosystems Dynamics Research, generates progenitor cells needed to build vital connective tissues for miniature livers, stomachs, esophagi and lungs.
For more than a decade, researchers worldwide have been getting better at coaxing stem cells to grow into tiny, simple versions of human organs—called organoids—that can mimic the key functions of everything from the brain to the colon. Now, scientists report tackling another challenge, figuring out how to make crucial cell types that are currently missing in many organoids. This will help make the next generation of organoids significantly more complex and realistic.
As a result, these improved organoids will become even more useful for studying disease and ultimately for making tissue that could be transplanted into patients for regenerative medicine.
Details were published online Aug. 17, 2022, in the journal Nature Protocols.
“These findings represent a significant step forward for next-generation organoids,” says Aaron Zorn, PhD, Director, Center for Stem Cell and Organoid Medicine (CuSTOM) at Cincinnati Children’s and senior co-author of the new paper.
Baking cakes with more layers
Baking a wedding cake takes more than assembling the ingredients along the kitchen counter. It takes more than mixing those ingredients into a good batter. Creating a multi-tiered masterpiece involves even more complexity and skill. Currently, organoid technology is closer to making tasty cupcakes.
“Most of the advances in organoid development so far have concentrated on the molecular signals needed to guide stem cells into specific epithelial cell types that form the walls, linings and glands that organs need to perform their functions, such as stomach cells that actually secrete digestive acids,” Zorn says. “This new protocol can generate a variety of smooth muscle and fibroblast cells that have been missing from the mix.”
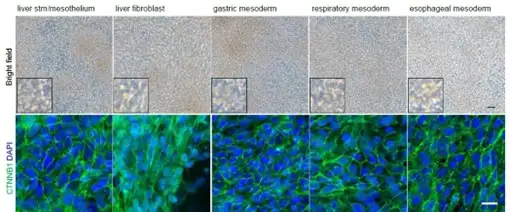
The research team, spearheaded by first author Keishi Kishimoto, PhD, a RIKEN-CuSTOM collaborative exchange scientist, used single-cell genetic analysis of mouse embryos to track down a complex set of molecular signals that direct precursor cells to form mesenchyme cells in different organs. Using this information, the team devised a 32-step protocol which over a week was able to cox human stem cells into five distinct mesenchyme progenitor populations. By activating or inhibiting four key cell signaling pathways—WNT, BMP, RA or HH—the team generated the liver septum, liver fibroblasts, gastric mesoderm, esophageal mesoderm, and respiratory mesoderm.
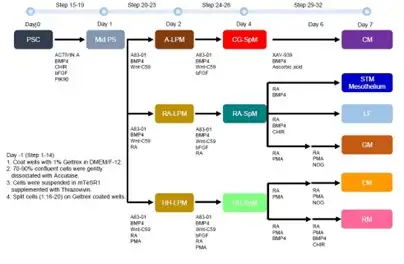
How Scientists Can Use This Discovery
This study details a protocol that was used by scientists at Cincinnati Children’s to produce the most complex stomach organoids grown to date. Details of that work appeared in December 2021 in Cell Stem Cell.
Having more complex organoids allows scientists to learn even more about how diseases occur and how human systems may respond to potential new medications.
“One of the most obvious applications is as platform to study the mechanisms of human splanchnic mesenchyme development, which is virtually unexplored,” Kishimoto says. “For example, it is possible to use our system to examine the molecular mechanisms by which these organ-specific mesenchyme progenitors differentiate into mature mesenchymal cell types such as fibroblasts, smooth muscle.”
Meanwhile, the more complex organoids can help experts conduct large-scale genetic studies to determine when, where and how organ formation goes wrong during human development, a line of research that cannot be studied effectively without organoids.
About the Study
In addition to Kishimoto and Zorn, co-authors from Cincinnati Children’s include Kentaro Iwasawa, MD, Alice Sorel, BS, Carlos Ferran-Heredia, MD/PhD student, Lu Han, PhD, James Wells, PhD, and Takanori Takebe, MD, PhD.
Funding for the work includes grants from the National Institutes of Health (P01HD093363), the NIH Director’s New Innovator Award (DP2 DK128799-01), a Cincinnati Children’s “Pursuing our Potential Together Award”, the New York Stem Cell Foundation, the Uehara Memorial Foundation, and the Japan Society for the Promotion of Science Overseas.
| Original title: | Directed differentiation of human pluripotent stem cells into diverse organ-specific mesenchyme of the digestive and respiratory systems |
| Published in: | Nature Protocols |
| Publish date: | Aug. 17, 2022 |
Research By