When Every Breath You Make Takes a Toll
Research By: Brian Varisco, MD
Post Date: January 13, 2022 | Publish Date:
COPD, Genetic Emphysema and the Potential for Antibodies to Halt Disease Progression
With each breath, the lung exchanges oxygen for carbon dioxide. Delicate and complex structures called alveoli mediate this process, and the destruction of these alveoli is termed emphysema.
Chymotrypsin-like elastase 1 (CELA1 ) is a naturally occurring protease that remodels the alveoli in response to strain and optimizes lung function, but its continued activity in emphysema leads to alveolar destruction and reduced lung function. Cincinnati Children’s physician researcher, Brian Varisco, MD, has developed a monoclonal antibody (KF4) against CELA1 that prevents alveolar loss in models of chronic obstructive pulmonary disease (COPD) and alpha-1 antitrypsin deficient emphysema—the most common genetic cause of emphysema.
One of the top causes of death in the United States and worldwide, COPD is characterized by two problems: mucinous obstruction of the conducting airways that causes airflow blockage, and emphysema. COPD makes breathing difficult for the 16 million Americans who have this disease. Patients with moderate to severe COPD also are at high risk for hospitalization from infection by COVID19.
Current treatment options for COPD and emphysema manage symptoms and are intended to prevent exacerbations, but so far there are no cures for emphysema in COPD.

Alpha-1 antitrypsin (AAT) neutralizes CELA1, and CELA1 is required for emphysema in models of AAT-deficient emphysema. By neutralizing CELA1, the antibody KF4 may be able to preserve lung function in patients with AAT deficiency. Alone or in combination with replacement therapy, KF4 may improve the quality of life and reduce mortality for patients with this genetic condition.
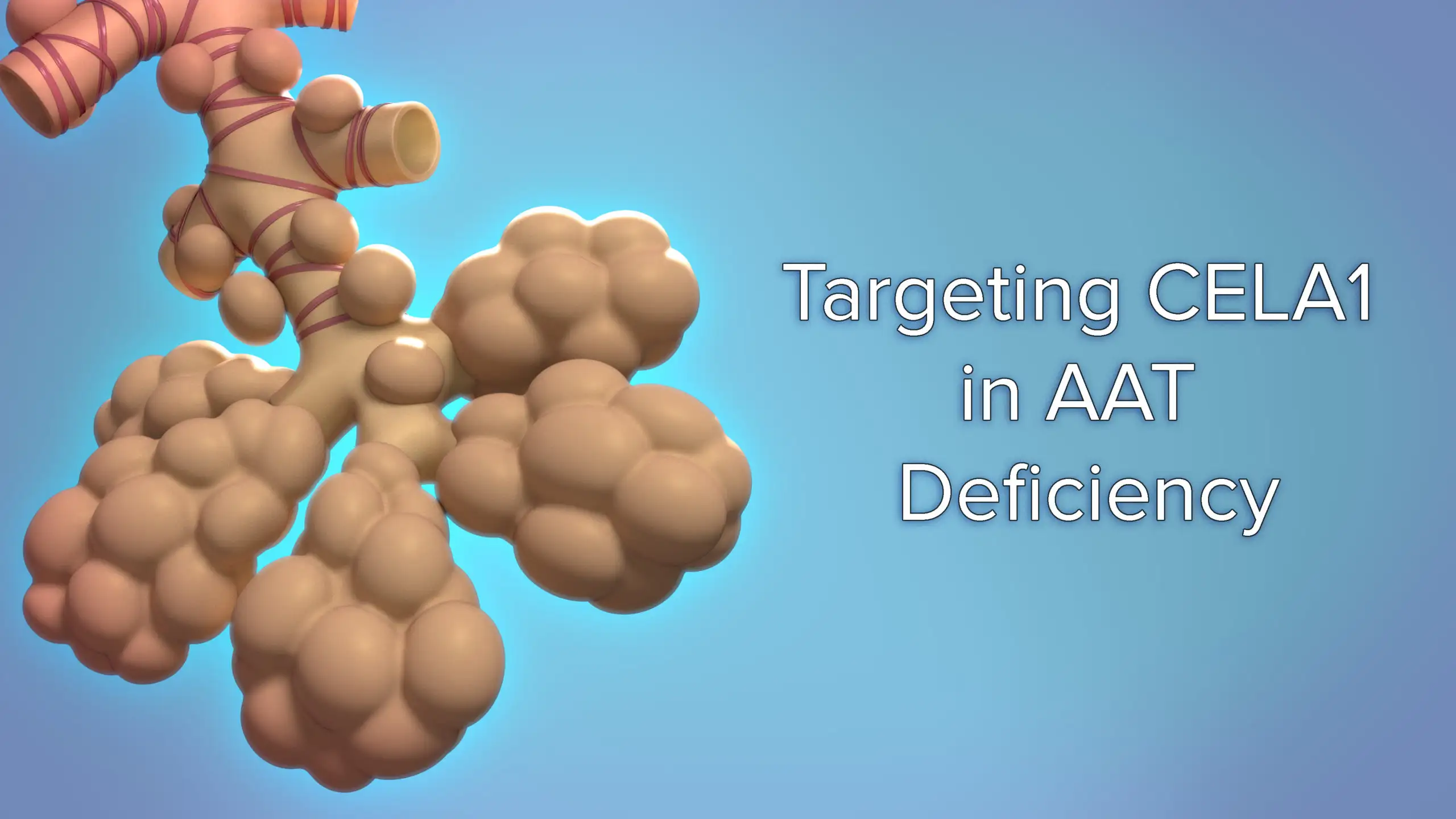
These video animations show the role that anti-CELA1 antibodies can play in mitigating disease progression. Varisco continues to collect efficacy data for KF4 in both wild type and AAT-knockout murine models.
Acknowledgments
Varisco’s research on the role of CELA1 has been supported by the National Heart, Lung and Blood Institute, the Alpha-1 Antitrypsin Foundation, the Parker B. Francis Family Foundation, the Cincinnati Children’s Research Foundation, and the Cincinnati Children’s Innovation Fund.
Credits
Media Lab at Cincinnati Children’s (@CincyKidsMedArt)
Animation: Jeff Cimprich, Matt Nelson, and Julia Bendon
Media Lab Direction: Ken Tegtmeyer, MD (@pccm_doc) and Ryan Moore, MD (@ramooremd)
Content Experts: Brian Varisco, MD (@BrianVarisco) and Eileen Yeoh (@CincyKidsInvent)
Voice Over: Carla Johnson
Learn more
For more information about the status of these and other technologies available for licensing, please visit Innovation.CincinnatiChildrens.org.
Research By