Study Decodes Critical Cell Signals Needed for Successful Pregnancy
Research By: Xiaofei Sun, PhD | Sudhansu K. Dey, PhD
Post Date: September 13, 2021 | Publish Date: Sept. 13, 2021
In mice, a small group of invasive cells that help establish blood supply from the placenta to the fetus depend on signals mediated by the gene Vangl2, according to experts at Cincinnati Children’s
In the complex journey of pregnancy, successful fetal development depends a great deal on how well the floating early-stage embryo sticks its landing against the inner lining of the uterus. Solid attachment assures a healthy blood supply while misguided or weak attachment can lead to miscarriage.
Now, experts at Cincinnati Children’s have discovered a crucial set of cellular signals that, when functioning correctly, helps guide the initial connection process, called placentation. Their findings were published online Sept. 13, 2021, in PNAS.
Sudhansu K. Dey, PhD, Director of the Division of Reproductive Sciences at Cincinnati Children’s has devoted years to understanding the complex initial moments of early-stage pregnancy. He calls these findings a “major breakthrough.”
“What we found here is that if a small group of placental cells doesn’t function properly, the feto-placental development undergoes demise,” Dey says.
Dey’s team at Cincinnati Children’s included Yeon Sun Kim, PhD, Yingju Li, PhD, Jia Yuan, PhD, and co-corresponding author Xiaofei Sun, PhD. The study also includes contributions from a collaborator in France, Jean-Paul Borg, PharmD, PhD, of Aix-Marseille University.
Completing a safe landing
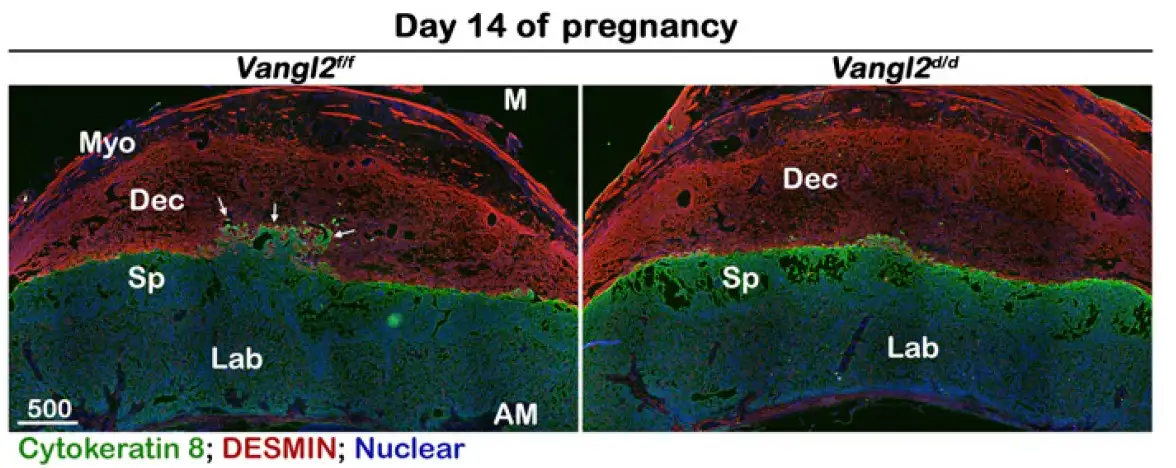
Dey and colleagues report that once the trophoblast lands on the decidua layer of the uterus, a group of spiral artery trophoblast giant cells (SpA-TGCs), invade the maternal decidua to establish blood supply for the fast-growing embryo. However, if those invading cells lack the gene Vangl2, they fail to penetrate deeply enough, and with weakened blood supply the fetus fails to grow, resulting ultimately in miscarriage.
The research team further found that the loss of Vangl2 specifically disrupts the balance of planar cell polarity (PCP) signaling and endocannabinoid signals that guide the trophoblast’s migration and its coupling with the decidua.
These findings shed light on the importance of cell polarity for successful placentation and healthy pregnancy, the co-authors say.
Is intervention possible?
Several more research steps are needed to determine if this discovery can lead to any treatments to reduce the risk of miscarriage.
Scientists still need to confirm that the process observed in mice also occurs in human pregnancy. With many suspected causes of miscarriage, it remains unclear how many occur because of this cause of poor placentation.
Preventing poor placentation would likely require establishing a testing protocol to find women with gene variants similar to the mice in the study, and developing a small-molecule drug that could influence cell polarity at the critical time, says co-author Xiaofei Sun.
“We are focusing on the downstream targets of this signaling complex, so that we may be able to better target this form of compromised pregnancy,” Sun says.
That work will take several years. In the meantime, women who want to become pregnant can help minimize the risks of early-stage signal disruptions by maintaining as healthy a lifestyle as they can, Dey says.
For example, marijuana use during pregnancy is known to increase the risk of miscarriage. Tobacco use should be avoided and alcohol use minimized. Communities also can help by reducing the ongoing barriers women in minority and low-income populations face in seeking affordable prenatal care.
About this study
This work was supported in part by grants from the National Institutes of Health (HD103475 and HD068524)
| Original title: | Cannabinoid and planar cell polarity signaling converges to direct placentation |
| Published in: | PNAS |
| Publish date: | Sept. 13, 2021 |
Research By








