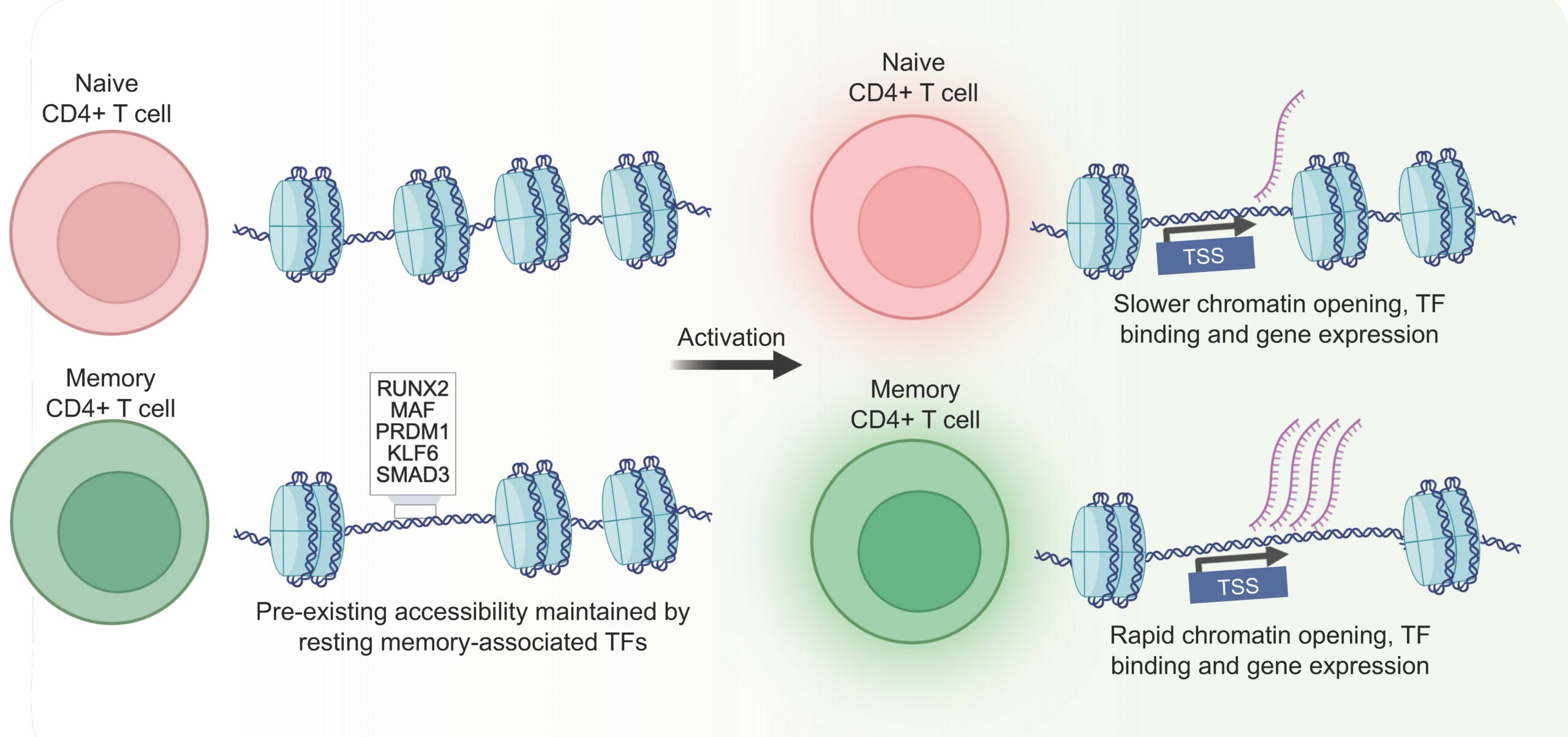
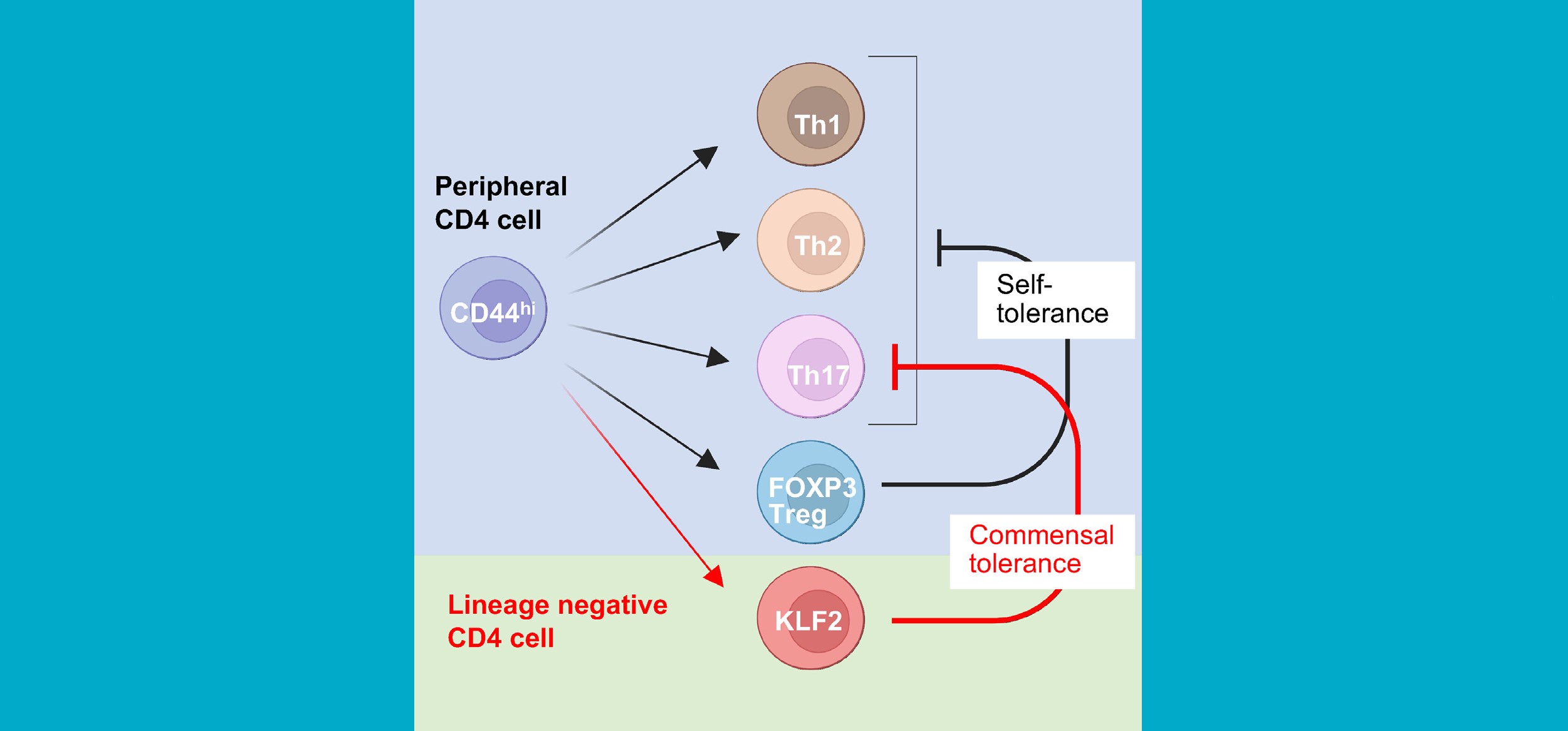
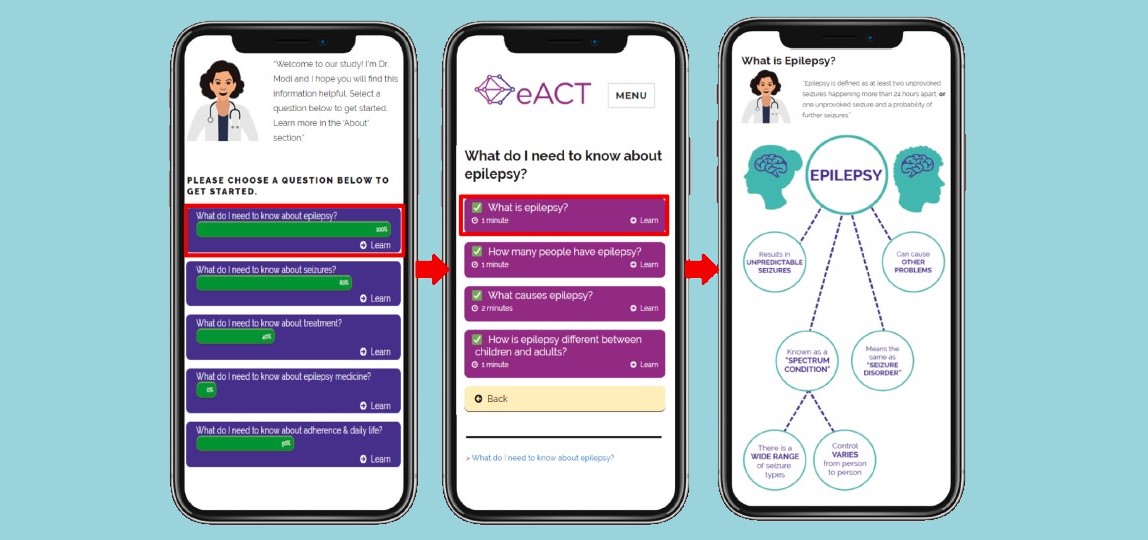
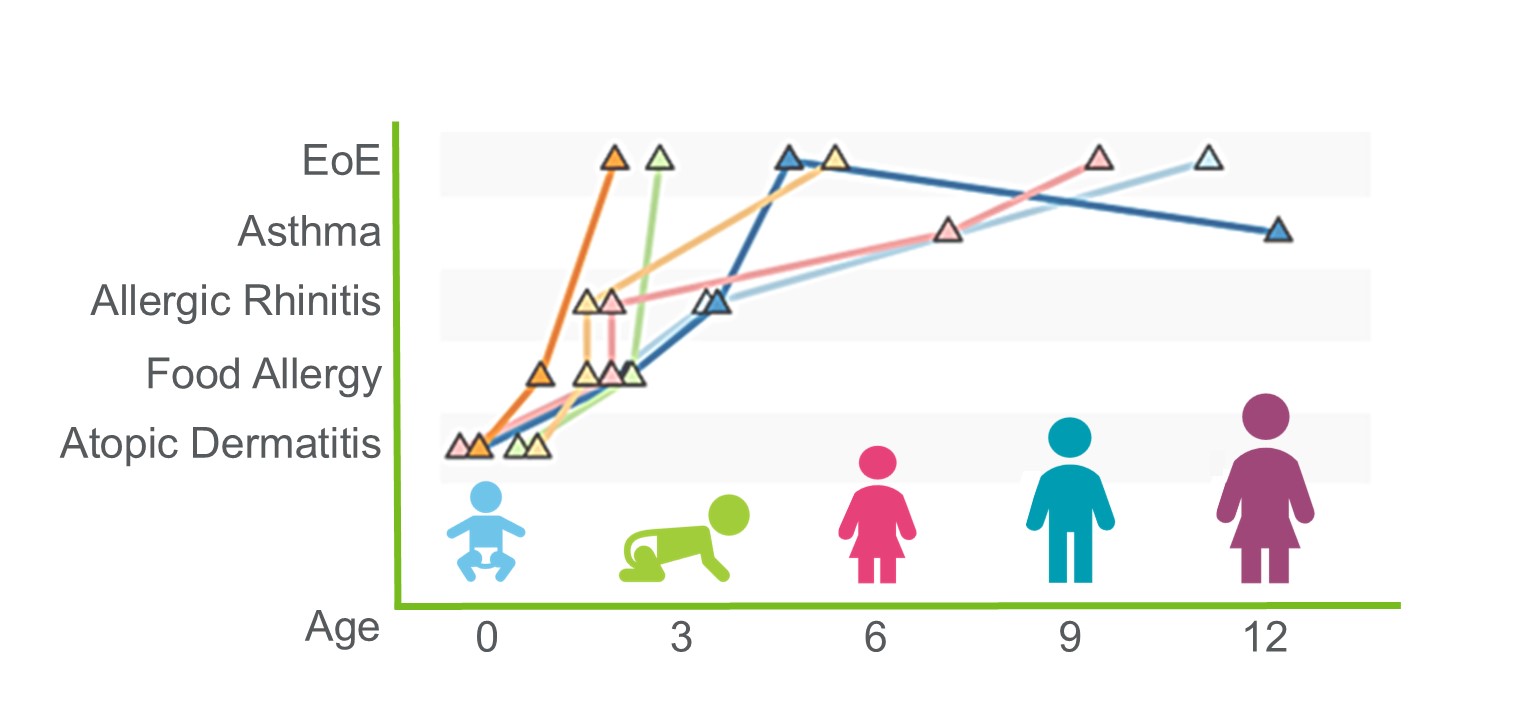
Progress Against the COVID-19 ‘Cytokine Storm’
Post Date: July 15, 2020 | Publish Date:
A study published June 24, 2020, in Lancet Rheumatology reports some of the best information to date on the use of the drug tocilizumab as a tool to control immune system overreaction to the novel coronavirus, according to a commentary written by Grant Schulert, MD, PhD, Division of Rheumatology.
The retrospective, observational cohort study led by a group of scientists in Italy reported outcomes for 544 adult patients who had severe COVID-19 pneumonia.
Of the patients who needed mechanical ventilation, 20% of those receiving standard care died, compared to 7% of those treated with tocilizumab.
“This study adds key new information to our understanding of tocilizumab in COVID-19. Previous studies of tocilizumab in COVID-19 have largely been encouraging, but either did not have a matched comparator group, did not match to contemporaneous controls, or were small and probably underpowered for key safety and efficacy outcomes,” Schulert wrote in an accompanying commentary.
“This study provides strong evidence that tocilizumab might prevent intubation and death in adults with severe COVID-19 pneumonia. These findings are also in agreement with emerging evidence that, in the setting of COVID-19-induced cytokine storm, immunosuppressive treatments might be most helpful earlier in the disease: after the onset of severe disease
but before florid respiratory failure. ”
“The precise group of patients who might benefit from tocilizumab and the optimal biomarkers for identifying cytokine storm in the setting of COVID-19 remain unknown. The most crucial question concerns the relative utility of tocilizumab treatment versus other non-specific immunomodulatory agents (including corticosteroids) and other cytokine-directed therapies, which is the focus of multiple ongoing randomized trials.”
More COVID-19 Science News
Moderna’s mRNA vaccine against the novel coronavirus induced anti–SARS-CoV-2 immune responses in all participants, without generating any trial-limiting safety concerns, according to this study in The New England Journal of Medicine.
Tracking circulating follicular helper T cell (cTFH) response may serve as a biomarker of potency for S-based vaccines entering the clinic, according to this study in Nature Medicine.
This paper, published in the Journal of Medical Virology, updates the epidemiological characteristics of COVID-19 in children.
HOW YOU CAN TRACK NEWS ABOUT COVID-19
COVID-19 Watcher: A pandemic data tracker created at Cincinnati Children’s and the University of Cincinnati that allows direct comparisons of city- and state-level activity
COVIDview: A Weekly Surveillance Summary of U.S. COVID-19 Activity from the CDC
COVID-19 projections assuming full social distancing through May 2020
Pandemic tracker from Worldometer
Pandemic tracking dashboard from Johns Hopkins University
Ohio Department of Health: COVID-19 dashboard
Guidance for care providers from Cincinnati Children’s
A frequently updated collection of pediatric COVID-19 papers can be found here