How a Fat Cell’s Immune Response Makes Obesity Worse
Research By: Senad Divanovic, PhD
Post Date: June 2, 2020 | Publish Date: June 2, 2020
Obesity has become an unabated pandemic. For over half a billion individuals, the accumulation of fat has led to life-shortening health consequences such as type II diabetes and non-alcoholic fatty liver disease (NAFLD).
Now, a team of scientists at Cincinnati Children’s and the University of Cincinnati College of Medicine has identified for the first time that adipocytes—the cells that compose the core of fat—can unlock a dormant inflammatory potential that may contribute to obesity-associated metabolic disruption. Their findings were published online June 2, 2020, in Nature Communications.
The study shows that for many people with obesity, a complex chain-reaction of signals within their fat appears to set them up for poor outcomes from several diseases. It also reveals an unappreciated mechanism driving the relationship between obesity and dysregulated immunity.
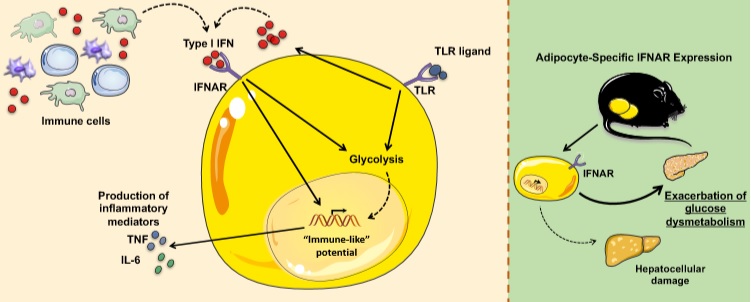
The team reports that type I interferons, a class of substances produced by adipocytes in addition to immune cells, drive a constant low-level, chronic immune response that amplifies “vigor” to a cycle of inflammation within white adipose tissue (WAT). More commonly known as white fat, this is the type of fat that expands to form most of the unwanted bulges around our thighs, arms and bellies.
This inflammation, in turn, appears to drive a cascade of cellular responses that promotes obesity-related disease, especially type 2 diabetes and NAFLD.
“Our novel study reveals how Type I Interferon sensing by adipocytes uncovers their dormant inflammatory potential and exacerbates obesity-associated metabolic derangements. Further, our findings highlight a previously underappreciated role for adipocytes as a contributor to the overall inflammation in obesity,” says Senad Divanovic, PhD, corresponding author and a researcher in the Division of Immunobiology at Cincinnati Children’s.
Health risks of obesity include poor infection outcomes
Obesity affects more than 600 million people worldwide—and the US has the highest average adult body mass index (BMI) of all high-income countries. By 2030, roughly half of the U.S. population could become obese (Ward et al., 2019).
Long known as a major risk factor for type 2 diabetes, NAFLD, cardiovascular disease, and diverse cancers, obesity also has been linked with elevated susceptibility and risk of developing serious complications to infection (Falagas & Kompoti, 2006). Obesity was an independent risk factor for severity and mortality in the 2009 H1N1 influenza pandemic (Van Kerkhove et al., 2011), and is a risk for hospital admission and poor outcomes among those infected in the current COVID-19 pandemic (Gupta, Ghosh, Singh, & Misra, 2020).
Many people understand that obesity is much more complicated than simply eating too much or not getting enough exercise. Previous work has shown that obesity also reflects the outcome of various disruptions to how the body converts food into energy for our cells. However, only recently have scientists begun to suspect that these excess calories could reshape fat cell behavior to affect the immune system. So far, the mechanisms underlying this interaction have not been well defined (Milner & Beck, 2012).
Adipocytes revealed as new target for type 1 interferons
The new study shows how type 1 interferons operate along an axis of interaction with IFNa receptors (IFNAR) to trigger a vicious cycle of inflammation. Among the effects, changes in expression of several genes associated with inflammation, glycolysis and fatty acid production.
For example, mice fed an obesity-inducing diet displayed an augmented type I IFN signature including increases in Ifnb1, Ifnar1, Oas1a, and Isg15 gene expression, the team reported.
Importantly, many of the metabolic changes documented in mice were found to be conserved in human adipocytes.
This activity was unexpected, because until now most scientists have studied type 1 interferons in relation to viral infections and immune cell function, including modulation of immune cell metabolic processes and production of immune mediators in this context.
“Our observations suggest that the type I Interferon axis can alter adipocyte core inflammatory programming to converge them closer to that of an inflammatory immune cell. Additionally, type I Interferons modify the metabolic circuit of adipocytes, which to our knowledge is the first depiction of immune-mediated modulation of adipocyte core metabolism,” Divanovic says.
New findings build upon bariatric surgery experiences
The study in Nature Communications reflects contributions from Divanovic, first author Calvin Chan—a member of the Medical Scientist Training Program at UC—and co-authors with expertise in obesity and metabolism, immunology, liver disease, bariatric surgery, genomics, biomedical informatics, organoid medicine and developmental biology.
Experts here been closely tracking how patients’ bodies change after receiving bariatric surgery. Work led at Cincinnati Children’s by Thomas Inge, MD–now at the University of Colorado–and colleagues has shown metabolic transformations that have gone beyond simply reducing food intake.
For example, in a number of cases, key signs of type 2 diabetes vanished independently of how much weight the patients lost.
“This study sheds light on one of the critical mechanisms that may have been at work in the metabolic changes occurring after bariatric surgical treatment,” the co-authors state.
What’s Next?
Further investigation continues into the specific mechanisms that type I Interferons employ to modify adipocyte core metabolism. In addition, researchers continue to study the full extent of how adipocytes can “mimic” inflammatory immune cell capabilities.
“These findings directly impact an extensive number of patients, both adult and pediatric,” Divanovic says. “Importantly, these findings are also suggestive of similar contribution of adipocytes in numerous diseases, including those associated with obesity or independent of obesity.”
Beyond diabetes and NAFLD, the interplay between obesity and the immune system appears to increase risk of preterm birth and may reduce the body’s ability to fight off infections—including viruses such as COVID-19.
| Original title: | Type I Interferon Sensing Unlocks Dormant Adipocyte Inflammatory Potential |
| Published in: | Nature Communications |
| Publish date: | June 2, 2020 |
Research By