FDA-Approved Drug Reverses Inflammation in Animal Model of Eosinophilic Esophagitis
Research By: Nurit Azouz, PhD | Marc Rothenberg, MD, PhD
Post Date: December 10, 2020 | Publish Date: May 27, 2020
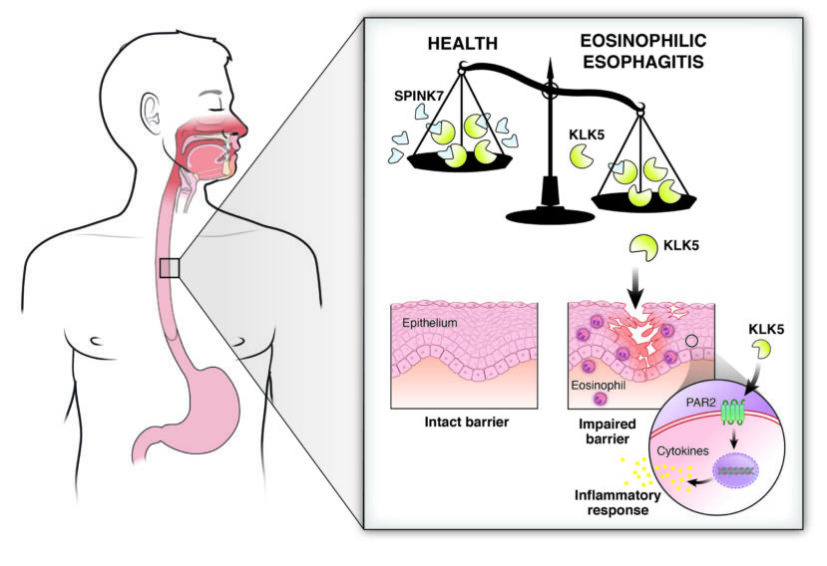
Scientists at Cincinnati Children’s have discovered that an imbalance of specific proteins, proteases, and protease inhibitors in cells lining the esophagus may cause inflammation and tissue damage in people with eosinophilic esophagitis (EoE).
This recently recognized chronic food allergy affects as many as 150,000 people in the U.S., many of whom are children. People with EoE experience difficult or painful swallowing, vomiting, and nutritional problems because of an accumulation of immune cells called eosinophils that cause scarring of the esophagus.
In people with active EoE, biopsies of esophageal tissues show a near-complete lack of the protease inhibitor, SPINK7, which normally damps down inflammation and helps preserve tissue structure. However, co-authors of a study published online May 27, 2020, in Science Translational Medicine, report that the biologic drug alpha-1 anti-trypsin (A1AT) reversed damaging inflammation in an animal model of EoE,
The study was led by first author Nurit Azouz, PhD, and senior author Marc Rothenberg, MD, PhD, Director of the Division of Allergy and Immunology.
How Does This Potential Treatment Help?
A1AT was approved to treat a genetic form of emphysema. So how does it help work against EoE?
Because foods we eat contain enzymes that can damage human tissue, the lining of the esophagus protects itself by producing its own enzyme inhibitors that prevent degradation of the offending proteins and thus protect the lining. Previously, Rothenberg and colleagues found that SPINK7 is a key contributor to this protective process and was nearly absent in the esophagi of adults and children with EoE. Without SPINK7, cultured esophageal cells lost their normal barrier function that ordinarily helps move food along the digestive tract. Tissues that did not express SPINK7 also produced high levels of chemical messengers called cytokines that attract and activate eosinophils and produce the same type of inflammation seen in allergic diseases.
In this study, researchers report finding a missing link that further explains how the process works. SPINK7 normally binds with the protease kallikrien 5, keeping it in check. Without enough SPINK7, kallikrien 5 becomes free to degrade tissues and start signals leading to inflammation.
“This is a breakthrough in understanding that the disease is caused by a balance between proteases and anti-proteases. If you shift this balance, you unleash the proteases,” Rothenberg says. “This causes the inflammation through the release of the inflammatory mediators that are associated with the eosinophils, which are the white blood cells that are characteristic of the disease.”
Biologic Replaces Role of SPINK7
A1AT is a natural inhibitor of tissue-damaging enzymes. To study its potential against EoE, Azouz and colleagues administered the drug to mice engineered to model the disease. They found that A1AT reversed the damaging inflammation seen in the esophagus.
The finding suggests that further investigation is warranted to determine whether the therapeutic may also benefit people with EoE. Current treatments for EoE include often-restrictive diets to avoid trigger foods, corticosteroids to relieve inflammatory symptoms, and sometimes surgical procedures to expand the esophagus.
“These findings highlight esophageal allergy as a protease-mediated disease. We suggest that disarming of proteases by delivering protease inhibitors has the potential to be therapeutic in allergic disease,” says Azouz.
Contributing co-authors from Cincinnati Children’s also include: Andrea Klingler, Purnima Pathre, John Besse, Netali Ben Baruch-Morgenstern, Adina Ballaban, Garrett Osswald, Michael Brusilovsky, Jeff Habel, Julie Caldwell, Mario Ynga-Durand, Pablo Abonia, Yueh-Chiang Hu, and Ting Wen.

Related Research
The new findings about A1AT build on a study led by Azouz, Rothenberg, and colleagues from multiple divisions at Cincinnati Children’s that was published in 2018.
Science Translational Medicine: The antiprotease SPINK7 serves as an inhibitory checkpoint for esophageal epithelial inflammatory responses
| Original title: | Functional role of kallikrein 5 and proteinase-activated receptor 2 in eosinophilic esophagitis |
| Published in: | Science Translational Medicine |
| Publish date: | May 27, 2020 |
Research By