Aruvant Presents Data on Improved Gene Therapy for Sickle Cell Disease
Post Date: December 8, 2020 | Publish Date:
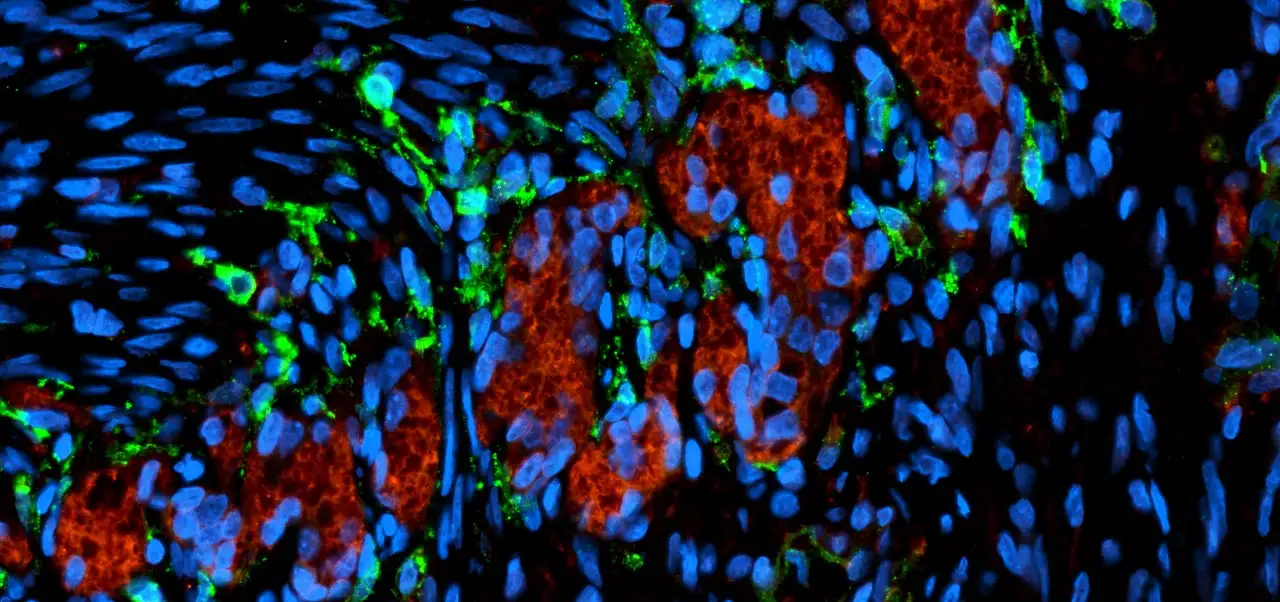
The third patient to participate in a clinical trial of a sickle cell gene therapy showed encouraging response to a treatment produced through an enhanced manufacturing process, according to data presented Dec. 7, 2020, at the 62nd American Society of Hematology (ASH) Annual Meeting.
The treatment, now being developed by Aruvant Sciences, a spin-off company co-launched by Cincinnati Children’s, is being studied in a phase 1/2 clincal trial called MOMENTUM.
“ARU-1801 demonstrated clinically meaningful long-term reductions in sickle cell disease burden,” said Michael Grimley, MD, Medical Director of the Division of Bone Marrow Transplantation and Immune Deficiency at the Cincinnati Children’s. “The potentially curative efficacy seen from the new manufacturing process and three year durability demonstrated by the first treated patient show that ARU-1801, given with only reduced intensity conditioning, can be an important and more tolerable treatment option for patients with severe SCD.”
The company used a new manufacturing process to treat “patient 3” in the study. Outcomes have included:
- Fetal hemoglobin of 38 percent at nine months.
- F cells (red blood cells that contain fetal hemoglobin) were 71 percent at six months and are expected to rise as transfused blood recedes.
- F-retics (new red blood cells that contain fetal hemoglobin) were 92 percent at six months.
- Normalization of total hemoglobin levels and measures of hemolysis.
- No vaso-occlusive episodes (“VOEs”) through nine months.
This Phase 1/2 study is currently enrolling participants, and information may be found at www.momentumtrials.com.
See more of the information presented at the conference.