2nd FLASH Proton Therapy Clinical Trial Begins
Research By: John Perentesis, MD
Post Date: March 28, 2023 | Publish Date:
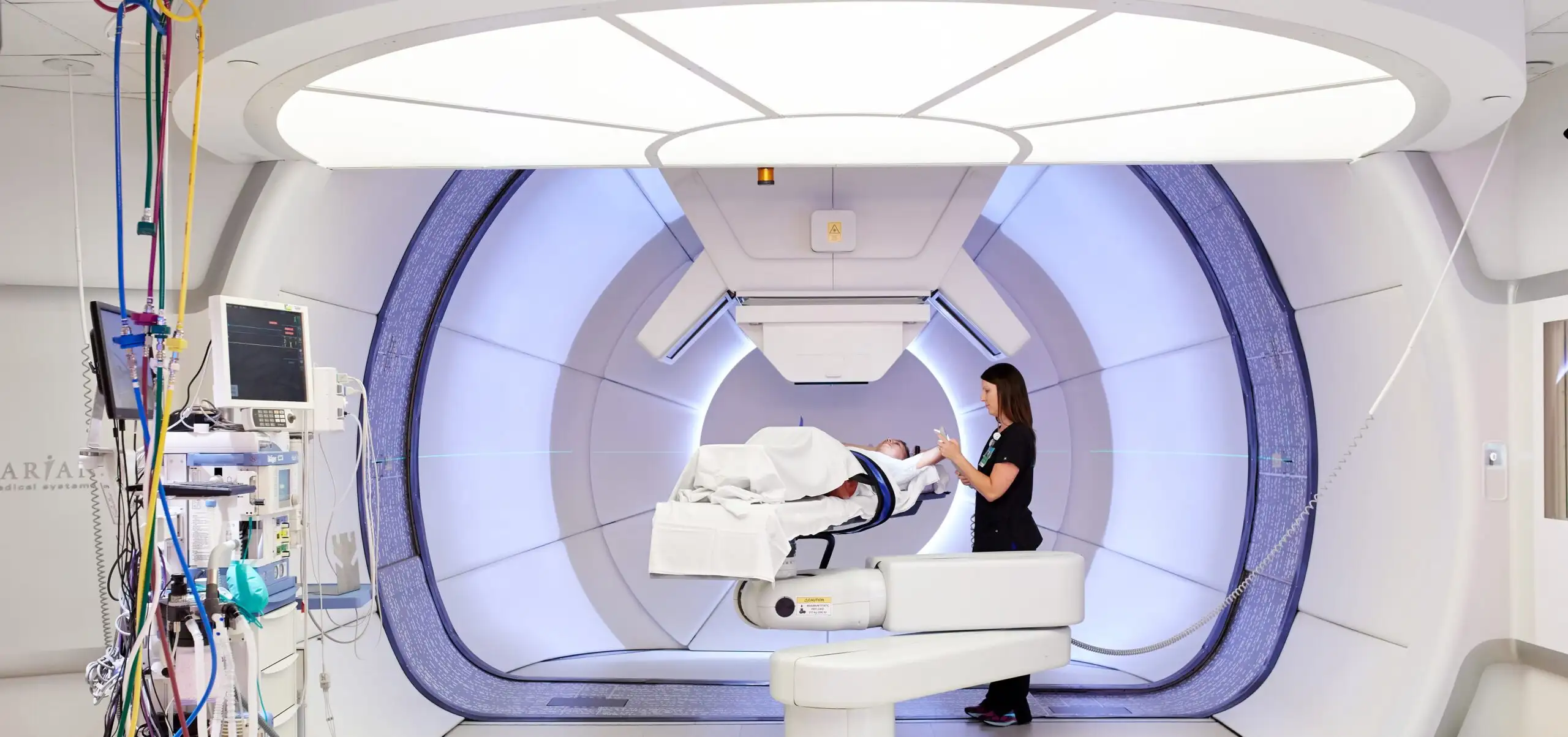
FAST-02 trial will focus on people whose metastatic cancer has spread to their chest bones
Treatment of the first participant in a clinical trial of FLASH proton therapy for cancers in the bones of the chest has been completed at the Cincinnati Children’s/University of Cincinnati Medical Center Proton Therapy Center.
FLASH is an investigational mode of radiation therapy delivery that can be administered to a patient in less than 1 second. Research in the laboratory suggests that FLASH may result in fewer side effects than standard forms of radiation delivery, and the clinical trial is testing the feasibility of FLASH to treat cancer sufferers.
The new study, called FAST-02, marks a significant achievement for the Proton Therapy Center in Cincinnati, which continues to lead the way in global research in the field of FLASH proton therapy.
Cincinnati Children’s researchers are collaborating with University of Cincinnati researchers to establish the efficacy of ultra-high dose rate proton therapy in treating painful bone metastases in the chest. The FAST-02 study is sponsored by Varian, a Siemens Healthineers company.
“This trial of an ultra-high dose rate of proton therapy is just one of our efforts to develop new and innovative cancer treatments,” said John Perentesis, MD, who is research director of the Proton Therapy Center and director of oncology and cancer programs at Cincinnati Children’s. “Our goal is to establish a foundation for future potential trials in brain tumors, sarcomas, lymphomas, lung cancer, and other malignancies, and we are excited about the impact this research might have on cancer care.”
Read more about the new clinical trial in our Newsroom
Watch Dr. Perentesis discuss the Proton Therapy Center on Spectrum 1
Read more about the Proton Therapy Center
Research By

Dr. Perentesis leads an active laboratory research program in the molecular etiology and pharmacogenetics of childhood leukemia and other cancers, with a focus on Down syndrome-related leukemias.







